Calculate the standard enthalpies of formation of (a) KClO 3 (s) from the enthalpy of formation of
Question:
Calculate the standard enthalpies of formation of
(a) KClO3(s) from the enthalpy of formation of KCl,
(b) NaHCO3(s) from the enthalpies of formation of CO2 and NaOH together with the following information:
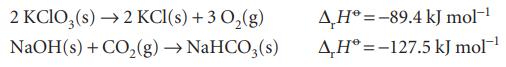
Transcribed Image Text:
2 KClO3(s) → 2 KCl(s) + 3 O₂(g) NaOH(s) + CO₂(g) → NaHCO3(s) AH-89.4 kJ mol-¹ AH-127.5 kJ mol-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
Solution a From the data given enthalpy of formation of ClO3 is 9626 kJ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The standard enthalpies of formation of gaseous propyne (C3H4), propylene (C3H6), and propane (C3H8) are + 185.4, +20.4, and -103.8 kJ/mol, respectively. (a) Calculate the heat evolved per mole on...
-
`The standard enthalpies of formation of ions in aqueous solutions are obtained by arbitrarily assigning a value of zero to H+ ions; that is, Hf [H+(aq)] = 0. (a) For the following reaction ...
-
The standard enthalpies of formation of S(g), F(g), SF4(g), and SF6(g) are 1278.8 kJ/ mol, 179.0 kJ/ mol, 775 kJ/ mol, and 1209 kJ/ mol, respectively. a. Use these data to estimate the energy of an...
-
What is a personal balance sheet? Name three classifications of assets. Briefly define and give examples of each.
-
Record in T accounts the following stock transactions of Pigua Company, which represent all of the companys treasury stock transactions during 2010: May 5 Purchased 1,600 shares of its own $2 par...
-
The codon change (Gly-12 to Val-12) in human H-ras that converts it to oncogenic H-ras has been associated with many types of cancers. For this reason, researchers would like to develop drugs to...
-
Gift Shop UBIT. A local exempt organization that trains at-risk youth for employment has an annual operating budget of $300,000, which includes revenue from operating a gift shop in a nearby hotel...
-
1. A Pleasant Evening with Delta "functions" Let's define the Dirac delta "function" 8(x) by the property for "well-behaved" functions f. [ (x)6(x) dx = (0), (a) Consider the family of box functions....
-
Capstone Case: Sunrise Bakery Expansion The Sunrise Bakery Corporation was originally founded in Houston, TX in 1991 by Griffin Harris, who currently serves as the company's Chief Executive Officer....
-
Show that the following functions have exact differentials: (a) X 2 y + 3y 2 , (b) X cos xy, (c) X 3 y 2 , (d) T(t + e s ) + s.
-
Calculate the standard enthalpy of solution of AgCl(s) in water from the enthalpies of formation of the solid and the aqueous ions.
-
The comparative balance sheets for Holders Company show these changes in noncash current asset accounts: accounts receivable decrease $80,000, prepaid expenses increase $18,000, and inventories...
-
Identify the best answer for each of the following: 1. Which of the following is not considered to be part of the basic financial statements? a. Managements Discussion and Analysis. b....
-
How should a government determine the appropriate balance to report for each of the three components of net assets of an Enterprise Fund?
-
Which of the following statements regarding the reporting of individual fund financial statements is true? a. Detailed budgetary information for the General Fund may be included as an individual fund...
-
GAAP require fiduciary activities to be reported a. in the fund financial statements by fund type. b. in the fund financial statements by major fund and in the government-wide financial statements as...
-
What is the accounting equation for a proprietary fund? What are the three components of net assets? Explain the nature of each component.
-
Litigation in which one company sues another often involves costs for lawyers and other court costs literally amounting to hundreds of millions of dollars per case. What does the concept of an...
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
What are the advantages of working with normalized wavefunctions?
-
Functions of the form sin(nx/L) can be used to model the wavefunctions of electrons in a carbon nanotube of length L. Show that the wavefunctions sin(nx/L) and sin(mx/L), where nm, are orthogonal for...
-
A particle free to move along one dimension x (with 0x
-
Describe all the TATA MOTORS "types of retailers" and the distribution components that will be involved in the new global product or service. List any differences between the TATA global channels...
-
Provide an explanation as to why the bank of scotia has selected India for the expansion rather than other international markets that could have been pursued. What specifically makes it attractive...
-
How Social Media Plays a Part in Creating A Brand's "Celebrity" For 30 Points: Many companies have turned to social media to enhance their brand's "celebrity" status - maybe they use a celebrity...

Study smarter with the SolutionInn App


