Construct the electron density along the x-axis of a crystal given the following structure factors: h 0
Question:
Construct the electron density along the x-axis of a crystal given the following structure factors:
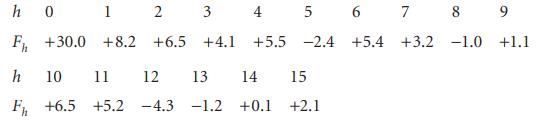
Transcribed Image Text:
h 0 F₁ +30.0 h 10 11 12 13 14 15 F₁ +6.5 +5.2 -4.3 -1.2 +0.1 +2.1 1 2 +8.2 +6.5 3 4 5 6 7 +4.1 8 9 +5.5 -2.4 +5.4 +3.2 -1.0 +1.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 86% (15 reviews)
Answer The electron density along the xaxis of a crystal is calcul...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A rod AB oriented along the x axis of the reference frame K moves in the positive direction of the x axis with a constant velocity v. The point A is the forward end of the rod, and the point B its...
-
The following images show contours of constant electron density for H 2 calculated using the methods described in Chapter 26. The values of electron density are (a) 0.10, (b) 0.15, (c) 0.20, (d)...
-
The random variable X has density function f given by (a) Determine K in terms of θ. (b) Find FX(b), the CDF of X. (c) Find E(X). (d) Suppose for OS vs 0. elsewhere.
-
Given the following set of slope staking notes: C Sta 51+00 50+00 49+00 L CX 33.4 F 9.1 33.6. F 10.3 35.4 C 5.5 0.0 F 3.5 R C 3.2 X C 4.1 30.2 0.0 20.0 Bases Base for cut=48 ft Base for fill= 40 ft s...
-
ACNielsen is a New Yorkbased leading global provider of marketing research information services, analytical systems and tools, and professional client service. A recent issue of its magazine...
-
Each of the gears A and B has a weight of 5 lb and a radius of gyration of 4 in., while gear C has a weight of 25 lb and a radius of gyration of 7.5 in. A couple M of magnitude 6.75 lb ??ft is...
-
An OEM assembled drive assemblies for its pumps. The following photograph was taken on one of the manufactured drive cabinets. The power cable between the motor and the drive was grounded by...
-
TBN manufactures jumpers for retail stores. John, the accountant of TBN, gets concerned about TBNs standard costing system. The budgeted and actual amounts below for August 2019 were drawn from its...
-
At the hearing on 30 January 2017, none of the parties was able to produce a copy of the contract which was the subject of the dispute. I took all of the parties' oral evidence, and received their...
-
On January 1, 2012, Prim Valley Resorts acquired 70 percent interest in Silverstem Casinos for $592 million in cash and stock. $580 million $233 million $100 million Book Value of Silverstem Casinos...
-
The coordinates, in units of a, of the atoms in a body-centred cubic lattice are (0,0,0), (0,1,0), (0,0,1), (0,1,1), (1,0,0), (1,1,0), (1,0,1), and (1,1,1). Calculate the structure factors F hkl when...
-
Potassium nitrate crystals have orthorhombic unit cells of dimensions a = 542 pm, b = 917 pm, and c = 645 pm. Calculate the glancing angles for the (100), (010), and (111) reflections using CuK ...
-
The principal component of natural gas is methane (CH4). How many moles of CH4 are present in 144.36 g of methane?
-
Assume convergence of yields to the mid-point of 3.61% and 3.26%, what is the profit for a successful trading where the long position is a $1,000 face value bond?
-
! Required information Problem 6-4A & 6-5A (Algo) [The following information applies to the questions displayed below.] Joseph Farmer earned $126,600 in 2020 for a company in Kentucky. He is single...
-
I need a step by step on how to input this into QuickBooks Week Ending August 29, 2026 Because the Wreath workshop the previous week was such a success, Melanie is offering a Build Your Own Bouquet...
-
Financial Instruments Scenario Classification of investment Balance Sheet Presentation Initial measurement Subsequent Measurement An entity acquires debt securities that it will hold until...
-
Asset Efficiency Ratios 2020 2021 2022 Asset Turnover Ratio Sales Revenue / (note used operating revenue) 826,912,000 990,607,000 1,127,475,000 Average Total Assets 669,599,000 956,235,000...
-
Reticulocyte (immature red blood cell) lysates can be made devoid of mRNA by ribonuclease treatment followed by inactivation and removal of the ribonuclease. These lysates are now capable of...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
Write down the expression for the radial distribution function of a 3p electron in a hydrogenic atom and determine the radius at which the electron is most likely to be found.
-
What is the orbital angular momentum of an electron in the orbital? (a) 4d, (b) 2p, (c) 3p? Give the numbers of angular and radial nodes in each case.
-
Calculate the permitted values of j for (a) A p electron, (b) An h electron.
-
How did they find the over head rate percentages for current year and last year? A job order cost sheet for Cullumber Company is shown below. Job No. 92 For 2,000 Units Date Direct Materials Direct...
-
An all-equity firm is considering the purchase of a depreciable asset that costs $680,000 and would be fully depreciated over six years using the straight-line method. The asset is expected to...
-
A firm owns three department stores. Each store has a debt-equity ratio of 16 percent and makes interest payments of $44,000 at the end of each year. The cost of the firm's levered equity is 18...

Study smarter with the SolutionInn App


