Derive the following relation, for the internal pressure of a gas that obeys the RedlichKwong equation of
Question:
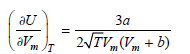
for the internal pressure of a gas that obeys the Redlich€“Kwong equation of state,
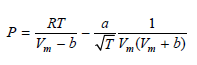
Transcribed Image Text:
За aU av т /T 2TV, т + b) || RT 1 V — в тиИ, + b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
The internal pressure o...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Derive an expression for the internal pressure of a gas that obeys the Bethelot equation of state, P = RT/V m b a TV 2 m.
-
A liquid-phase chemical reaction A ? B takes place in a well-stirred tank. The concentration of A in the feed is C A0 (mol/m 3 ), and that in the tank and outlet stream is C A (mol/m 3 ). Neither...
-
The first-order thermal decomposition of chlorocyclohexane is as follows: C 6 H 11 Cl(g) C 6 H 10 (g) + HCl(g). For a constant volume system, the following total pressure was measured as a function...
-
4. Finally, briefly discuss how you could use an empirical econometric model to assess the health and economic growth consequences of a 'fat tax'.
-
What do you think of this developer's attitude? Do you think he's unduly pessimistic or do you think there's merit to what he says?
-
Consider a molecule with fixed dipole \(\vec{d}\) in a time-dependent electric field \(\vec{E}(t)\). Calculate the Berry connection and Berry magnetic field in this case. Do we have a geometrical...
-
Identify objectives and solution features for the following products: a. An ATM. b. An accounting information system. c. A carwash. d. A car.
-
In general, how in your opinion did Netflix manage to put itself in the enviable position that it occupied at the outset of fiscal 2010? More specifically, how would you account for the suddenand...
-
Greenwood Company manufactures two products-15,000 units of Product Y and 7,000 units of Product Z. The company uses a plantwide overhead rate based on direct labor-hours. It is considering...
-
ADA Pharmaceutical Company produces three drugs: Diomycin, Homycin, and Addolin belonging to the analgesic (pain-killer) family of medication. Since its inception four years ago, ADA has used a...
-
Predict the product(s) for each of the following reactions: a. b. c. d. e. f. Ni Pd
-
What are some of the advantages of storing high-level radioactive waste at Yucca Mountain? What are some of the disadvantages?
-
What is the composition, in atom percent, of an alloy that consists of 97 wt% Fe and 3 wt% Si?
-
Gloria and Ed Phillips file a joint tax return and have an adjusted gross income (AGI) of $110,000 for 2020. Their son, Randall, began his first year of graduate school in 2019. The Phillips'...
-
You just got an offer for a bond issued by Coala Corporation. The bond pays annual coupons with a coupon rate of 0.035, while its yield to maturity is 0.1, the bond has 12 years to maturity what is...
-
The variance of stock A is 0.005 and the return is 0.115, while B has the same return but 0.15 as standard deviation of what is the coefficient of variation for stock A what is the coefficient of...
-
Bank A charges a 7.75 percent annual percentage rate and interest is due at the end of the year. Bank B charges a 7 percent annual percentage rate and interest must be paid monthly. What is the...
-
A bond with 9 years of life left and a coupon rate of 12% is selling at a price of INR 122.0067. The yield on the bond is 8.416%. What is the price value of 1bp?
-
According to the Original Tipping Page, www.tipping.org, it is appropriate to tip waiters and waitresses 15%20% of the pretax restaurant bill. The Leesebergs dinner costs $43.50 before tax, and the...
-
The power company must generate 100 kW in order to supply an industrial load with 94 kW through a transmission line with 0.09 resistance. If the load power factor is 0.83 lagging, find the...
-
Estimate the coefficients a and b in the Dieterici equation of state from the critical constants of xenon. Determine pressure exerted by 1.0 mol Xe when it is confined to 1.0 dm3 at 25C.
-
(a) Could 25 g of argon gas in a vessel of volume 1.5 dm3 exert a pressure of 2.0 bar at 30C if it behaved as a perfect gas? If not, what pressure would it exert? (b) What pressure would it exert if...
-
A perfect gas undergoes isothermal compression, which reduces its volume by 1.80 dm3. The final pressure and volume of the gas are 1.97 bar and 2.14 dm3, respectively. Calculate the original pressure...
-
Required: a. Prepare a cost of goods manufactured and sold statement. b. Prepare an income statement. Answer is not complete. Complete this question by entering your answers in the tabs below. COGM...
-
Syarikat Jaya uses normal costing for its costing system whereby the calculation of predetermined overhead rate based on direct labours costs. On 1st December 2022, the general ledger consists of the...
-
Ron owns 6 % of USAco, a U . S . corporation. Ron is a nonresident alien. USAco pays Ron a $ 1 , 2 0 0 dividend during the current year. Ron also purchases an unassignable bond issued by USAco, and...

Study smarter with the SolutionInn App


