Figure 5.8 shows the phase diagram for the ternary system NH 4 Cl/ (NH 4 ) 2
Question:
Figure 5.8 shows the phase diagram for the ternary system NH4Cl/ (NH4)2SO4/H2O at 25 °C. Identify the number of phases present for mixtures of compositions (i) (0.2, 0.4, 0.4), (ii) (0.4, 0.4, 0.2), (iii) (0.2, 0.1, 0.7), (iv) (0.4, 0.16, 0.44). The numbers are mole fractions of the three components in the order (NH4Cl,(NH4)2SO4,H2O).
Data in Figure 5.8
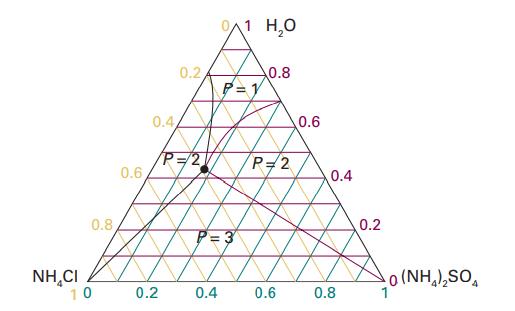
Transcribed Image Text:
NHẠCI 0.8 10 0.6 0.4 0.2 0.2/ P=2 0.4 0/1 H₂O P=1 0.8 P=2 0.6 0.6 0.4 0.8 0.2 0 (NH,),SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
At 25 C i For the mixture with mole fractions 02 04 04 there i...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Figure 5.2 shows the phase diagram for two partially miscible liquids, which can be taken to be that for water (A) and 2-methyl-1-propanol (B). Describe what will be observed when a mixture of...
-
The phase diagram for SO2 is shown here. (a) What does this diagram tell you about the enthalpy change in the reaction SO2(I) SO2(g)? (b) Calculate the equilibrium constant for this reaction at 100...
-
The phase diagram for neon is Temperature (K) Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point? (b) Over what pressure range will...
-
a. Find the probability of getting exactly 1 girl in 10 births. b. Find the probability of getting 1 or fewer girls in 10 births. c. Which probability is relevant for determining whether 1 is an...
-
On December 31, Year 1, Brown Brothers purchased machine A for $ 770,000 and machine B for $ 300,000. The machines are depreciated on the straight- line basis over 10 years with no salvage value ....
-
The following method was known to the ancient Greeks for computing square roots. Given a value x > 0 and a guess g for the square root, a better guess is (g + x/g) / 2. Write a recursive helper...
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
Common Chemical Companys management is evaluating its product mix in an attempt to maximize profits. For the past two years, Common has produced four products, and all have large markets in which to...
-
Peanut Company acquired 80 percent of Snoopy Company's outstanding common stock for $276,800 on January 1, 20X8, when the book value of Snoopy's net assets was equal to $346,000. Peanut uses the...
-
Although the vibrational degrees of freedom are generally not in the high-T limit, is the vibrational partition function evaluated by discrete summation?
-
Consider the phase diagram in Fig. 5.6, which represents a solidliquid equilibrium. Label all regions of the diagram according to the chemical species that exist in that region and their phases....
-
Find the relation between the standard and biological standard Gibbs energies of a reaction of the form A2B+2 H + (aq).
-
What limits are placed on the selection of a tax year of an S corporation? How do these limits differ from those applicable to C corporations and partnerships?
-
1. What is positive, what The Founder negative about Ray Kroc's character? 2. What ethical approach describes Kroc's activities? 3 What ethical position is taken by the McDonald's brothers? 4 What is...
-
The senior on a CPA firm's largest audit engagement received a request from the client's CFO for a copy of "any communications the firm has sent relating tointernal-control-relatedmatters identified...
-
In the "The Rationale of Cost Accounting" by R.S. Edwards the author writes about the problematic nature of costing overhead. In your own words, explain why costing overhead can be problematic and...
-
The velocity of an airplane flying into a headwind is given by v(t) = 30 (25-t2) mi/hr for 0 st4 hr. Assume that s(0) = 0. a. Determine the position function for Osts4. b. How far does the airplane...
-
Students in a particular class breathe a sigh of relief when they see that their professor does not have the red folder that he always carries into class when he administers pop quizzes. This CR by...
-
A control system is shown in Figure AP7.9. Sketch the root loci for the following transfer functions Gc(s): (a) Gc(s) = K (b) Gc(s) = K(s + 3) (c) (d) Figure AP7.9 A unity feedback control system. K...
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
Consider the equilibrium in the reaction 3O 2 (g) 2O 3 (g). Assume that H o R is independent of temperature. a. Without doing a calculation, predict whether the equilibrium position will shift...
-
Why does water have several different solid phases, but only one liquid and one gaseous phase?
-
Explain how the ideal gas law can be deduced for the measurements shown in Figures 1.5 and 1.8. Figure 1.5 Figure 1.8 0.1 L 2.5 2.0 1.5 0.2 L 1.0 0.3 L 0.4 L 0.5 L 0.6 L '0.5 -200 100 0 100 200 300...
-
1. One free t-shirt received at the time of enrollment. Each t-shirt costs Annie's $9.00. 2. One free ice cream flight per month. Each flight consists of two-ounce servings of four flavors. Annie's...
-
2. [This question is worth 3 points] Consider the series of cash flows shown in the figure below. Find the economic equivalence of this series (i.e. all of the individual cash flows) at the end of...
-
What are the challenges and opportunities associated with fostering a culture of integrity in rapidly evolving industries and digital ecosystems, and how can proactive integrity management strategies...

Study smarter with the SolutionInn App


