Figure 5.2 shows the phase diagram for two partially miscible liquids, which can be taken to be
Question:
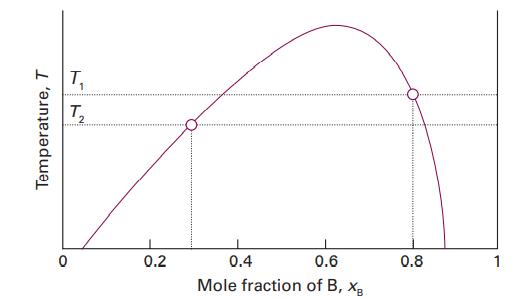
Figure 5.2 shows the phase diagram for two partially miscible liquids, which can be taken to be that for water (A) and 2-methyl-1-propanol (B). Describe what will be observed when a mixture of composition xB =0.8 is heated, at each stage giving the number, composition, and relative amounts of the phases present.
Data in Figure 5.2
Transcribed Image Text:
Temperature, T T₁ 1 T₂ 0.2 0.4 0.6 Mole fraction of B, XB 0.8 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
As the mixture is heated the following will be observed 1 At t...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Figure 5.8 shows the phase diagram for the ternary system NH 4 Cl/ (NH 4 ) 2 SO 4 /H 2 O at 25 C. Identify the number of phases present for mixtures of compositions (i) (0.2, 0.4, 0.4), (ii) (0.4,...
-
What steps can be taken to make controlling costs easier? Can these steps also be used to control other project parameters, such as scope?
-
What actions can be taken to reduce derived demand?
-
Khairul has been working with Tun Hamid Energy Bhd since 1 February 2009 as a Human Resource Executive. Beginning 1 January 2014, he was promoted as Human Resource Manager. Details of his...
-
The rules related to the capitalization of interest costs into the original carrying value of PPE are generally similar under U. S. GAP and IFRS. While there are some differences, most notably in how...
-
Reimplement the LabeledPoint class of Exercise E9.15 by storing the location in a java.awt.Point object. Your toString method should invoke the toString method of the Point class. Data from Exercise...
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
John Fuji (age 37) moved from California to Washington in December 2013. He lives at 468 Cameo Street, Yakima, WA 98901. Johns Social Security number is 571785974 and he is single. His earnings and...
-
Give any statement as an example to explain data manipulation language nature of SQL.?
-
A friend suggests that organizational behavior courses are useful only to people who will enter management careers. Discuss the accuracy of your friends statement.
-
Suggest an interpretation of the additional terms in extended versions of the DebyeHckel limiting law.
-
Show that the osmotic pressure of a real solution is given by V=RT ln aA. Go on to show that, provided the concentration of the solution is low, this expression takes the form V=RT[B] and hence that...
-
The time from acceptance to maturity on a $1,000,000 banker's acceptance is 120 days. The importer's bank's acceptance commission is 1.75 percent and the market rate for 120-day B/As is 5.75 percent....
-
5. A prime number is a number that is evenly disible by and itself. The prime numbers less than 100 are: 2 3 4 7 11 13 17 19 23 29 31 37 41 43 47 53 59 61 67 71 73 79 83 89 97 Choose a number at...
-
a. Define entrepreneurship and entrepreneurs. The students should provide 3 definitions of entrepreneurship and 2 definitions of an entrepreneur using relevant literature. They are required to...
-
The Journal Entry Application's (JEA) function is to create journal entries. The JEA receives notice from the Billing Process Application (BPA) that an invoice has been created. The JEA requests the...
-
The principle, humans cannot remove their basic emotions to make a determination of right and wrong. Does individual agree or disagree with this, explain why? If we adhere to the principle, how is...
-
The Nichol building at Carleton University was constructed as a space for students, faculty, staff, alumni, and the community to engage and find solutions to business challenges. The idea for the...
-
The top view of a high-performance jet aircraft is shown in Figure AP7.1(a) [20]. Sketch the root locus and determine the gain K so that the ζ of the complex poles near the jÏ-axis...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
One mole of H 2 O(l) is super cooled to 3.75C at 1 bar pressure. The freezing temperature of water at this pressure is 0.00C. The transformation H 2 O(l) H 2 O(s) is suddenly observed to occur. By...
-
An athlete at high performance inhales ~3.75 L of air at 1.0 atm and 298 K at a respiration rate of 32 breaths per minute. If the exhaled and inhaled air contain 15.3 and 20.9% by volume of oxygen,...
-
The temperature of 1.75 moles of an ideal gas increases from 10.2C to 48.6C as the gas is compressed adiabatically. Calculate q, w, U, and H for this process assuming that C V ,m = 3/2 R.
-
How do you decide upon your timeline? What are the key milestones? How do you ensure your timeline is done in a SMART (specific, measurable, attainable, relevant, and timebound) way?
-
With clear examples, compare and Contrast Constitutional Law and Administrative Law Discuss with case laws that defines the executive power?
-
Determine whether the following statements are true or false. Justify your answers. (a) The set Z7 = {0,1,2,3,4,5,6} is a group under multiplication modulo 7. (b) (c) The relation R on Z defined by...

Study smarter with the SolutionInn App


