Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
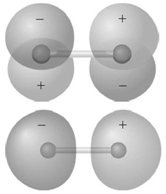
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
The top image has symmetry and a single node in ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
ed The Engine Guys produces specialized engines for "snow climber buses. The company's normal monthly production volume is 2,500 engines, whereas its monthly production capacity is 5,000 engines. The...
-
Consider the functions f (x) = sin x and f -1 (x) = arcsin x. (a) Use a graphing utility to graph the composite functions f o f 1 and f 1 o f. (b) Explain why the graphs in part (a) are not the graph...
-
List and briefly explain the different types of cash.
-
Of the following four research questions, which ones are appropriate for the chi-square test? a. What is the difference between the average scores of two math classes? b. What is the difference...
-
Daniel Grady is the financial advisor for a number of professional athletes. An analysis of the long-term goals for many of these athletes has resulted in a recommendation to purchase stocks with...
-
Milo Company manufactures beach umbrellas. The company is preparing detailed budgets for the third quarter and has assembled the following information to assist in the budget preparation: a. The...
-
Corporate ownership varies around the world. Historically, individuals owned the majority of shares in public corporations in the United States. In Germany and Japan, however, banks, other large...
-
Why are MOs on hetero-nuclear diatomics not labeled with g and u subscripts?
-
What is the justification for saying that, in expanding MOs in terms of AOs, the equality j (1) = c ij i (1) can in principle be satisfied?
-
The probability that an electronic component will fail in less than 1,000 hours of continuous use is 0.25 . Use the normal approximation to find the probability that among 200 such components fewer...
-
An organization's health and longevity depends on its ability to adapt and grow in ever changing environments. Boyne Mountain and Boyne Highlands have experienced their share of steady growth. Boyne...
-
Perform the complete the square operation for the expression 7p-10p-8
-
Revco Corporation purchases 35 percent of the voting stock of Ronco Pharmaceuticals on January 1, 2022, for $15 million in cash. Ronco's book value at the date of acquisition is $10 million....
-
Evaluate, correct to 2 decimal places When a = 3.43 b=1.55 c = 5.78 d = -0.26 ab-cd c2 d
-
1: Which do you prefer McGregor's Theory X or Theory Y? What does this mean about your personal management style and the type of person you are to work for? With? Pick two authors that we've read...
-
Find the sum. (-+}) 80 - 2.
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Given that the spacing of lines in the microwave spectrum of 27 Al 1 H is constant at 12.604 cm 1 , calculate the moment of inertia and bond length of the molecule (m( 27 Al) = 26.9815 u).
-
Pure rotational Raman spectra of gaseous C 6 H 6 and C 6 D 6 yield the following rotational constants: B(C 6 H 6 ) = 0.189 60 cm 1 , B(C 6 D 6 ) = 0.156 81 cm 1 . The moments of inertia of the...
-
The rotational constant of NH 3 is equivalent to 298 GHz. Compute the separation of the pure rotational spectrum lines in GHz, cm 1 , and mm, and show that the value of B is consistent with an N-H...
-
Reed Company had the following account balances for the current year. Prepare a multiple-step income statement. U Accounts Receivable $ 26,000 Cost of Goods Sold 63,400 Dividends 22,900 General and...
-
Welch Corporation just paid its annual dividend of $2.00 per share. The firm is expected to grow at a rate of 15 percent for the next three years and then at 6 percent per year thereafter. The...
-
Generous Limited has issued a callable bond at 5.35% coupon with 14 years left to maturity can be called in 4 years. The call premium is one year of coupon payments. It is offered for sale at...

Study smarter with the SolutionInn App


