Question: Predict whether LiH + 2 and NH 2 should be linear or bent based on the Walsh correlation diagram in Figure 24.11. Explain your answers.
Figure 24.11
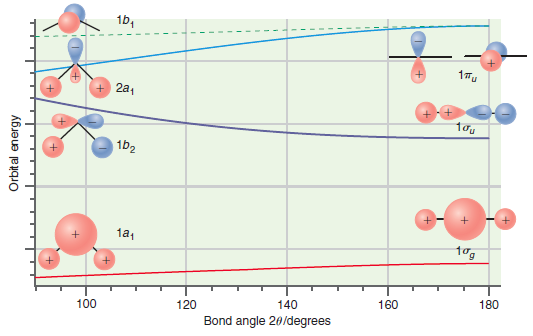
1b, + 2a, 1b2 1a1 tog 100 120 140 160 180 Bond angle 20/degrees Orbital energy
Step by Step Solution
3.59 Rating (167 Votes )
There are 3 Steps involved in it
The LiH 2 molecular ion has two valence electrons Th... View full answer

Get step-by-step solutions from verified subject matter experts


