Refer to the ternary phase diagram in Fig. 5D.4. How many phases are present, and what are
Question:
Refer to the ternary phase diagram in Fig. 5D.4. How many phases are present, and what are their compositions and relative abundances, in a mixture that contains 2.3 g of water, 9.2 g of chloroform, and 3.1 g of acetic acid? Describe what happens when (i) water, (iii) acetic acid is added to the mixture.
Data in Fig. 5D.4.
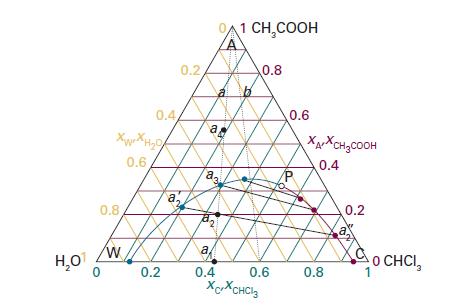
Transcribed Image Text:
H₂O Xuan Ho 0.6 0.8 0.4 W 0.2 a 20 0.2 a a. 23 a 0.4 1 CH₂COOH 0.8 0.6 XCXCHCl3 0.6 XAXCH₂COOH 0.4 0.8 0.2 O CHCI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
For the given mixture there are three phases present wat...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
A mixture that contains 46 wt% acetone (CH3COCH3), 27% acetic acid (CH3COOH), and 27% acetic anhydride [(CH3CO) 2O] is distilled at P = 1 atm. The feed enters the distillation column at T = 348 K at...
-
How many phases are present at each of the points marked in Fig. 4.1a? Data in Fig. 4.1a? Pressure (a) d b Temperature O a
-
A mixture that contains benzoic acid, 4-chloroaniline and naphthalene. The 4-chloroaniline is separated first by extraction with hydrochloric acid.Since no phenolic compound is present in this...
-
Consider the following velocity distribution curves A and B. a. If the plots represent the velocity distribution of 1.0 L of He(g) at STP versus 1.0 L of Cl2(g) at STP, which plot corresponds to each...
-
Does recognizing an impairment loss on a long- term operating asset have the same effect on the financial statements as recording depreciation expense and amortization expense? Explain.
-
Make a bar chart to plot the following data set. Label each bar. Provide a class BarChartViewer and a class BarChartComponent. Bridge Name Longest Span (ft) Golden Gate 4,200 Brooklyn 1,595 Delaware...
-
Maribel Baltazar was hired by clothing retail merchandiser Forever 21 in 2007. During the hiring process, Baltazar was given an 11-page document to sign, two pages of which contained an arbitration...
-
Refer to Golden Corporations financial statements and related information in Problem 16- 4A. In Problem 16- 4A, Golden Corp., a merchandiser, recently completed its 2013 operations. For the year, (1)...
-
what is Data Manipulation Language ? How is it related to DBMS ?
-
Crop-Quick Inc. replenishes its three distribution centers in Boston, Denver and Houston from its Las Vegas central supply warehouse. The distribution schedule for one of its products for the next...
-
Use mathematical software, a spreadsheet, or the Living graphs on the web site for this book to plot p A /p A * against x A with =2.5 by using eqn 5E.19 and then eqn 5E.20. Above what value of x A...
-
Could a regular tetrahedron be used to depict the properties of a four component system?
-
The Art Institute of Grosse Out Pointe has decided to evaluate pledge renewal rates for new donors. The director feels that the Institute will be on solid footing if at least 55 percent of last years...
-
The definition of a liability forms an important element of the International Accounting Standards Board's Framework for the Preparation and Presentation of Financial Statements which, in turn, forms...
-
What is the formula to calculate the current ratio? What does the current ratio measure? What is the formula to calculate the debt ratio? What does the debt ratio measure? When would you use each of...
-
Based on the COVID-19 event in Zambia, what issues were recorded within the country's main supply chain management? By identifying trends through examining the principles and standards, diversity,...
-
What public interest groups, aging services, or professional organizations have the most influence on political decisions related to long-term care? Why? What was the significance of the passage of...
-
On June 14, 2019, Montgomery Oil & Chemicals sold 185,000 gallons of oil and other lubricants to Qumicos Industriales Co. at a price of $1.90 per gallon. On August 23, 2019, 65,000 gallons were...
-
A unity feedback control system shown in Figure AP7.14 has the process Design a PID controller using Ziegler-Nichols methods. Determine the unit step response and the unit disturbance response. What...
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
Use the relation the cyclic rule and the van der Waals equation of state, to derive an equation for C P,m C V,m in terms of V m , T, and the gas constants R, a, and b. aV . Cr m = T
-
The amino acid glycine dimerizes to form the dipeptide glycylglycine according to the reaction 2Glycine(s): Glycylglycine(s) + H 2 O(l) Calculate ÎS, ÎS surr , and ÎS suniverse at T...
-
A 3.75 mole sample of an ideal gas with C V ,m = 3/2 R initially at a temperature T i = 298 K and P i = 1.00 bar is enclosed in an adiabatic piston and cylinder assembly. The gas is compressed by...
-
What are the challenges and also ethical issue related to the organization structure of the small scale medical clinic, give examples of challenges and also ethical issue and write down the reason.
-
a) The variables p and q are connected by the formula pea +48e9 +6. i) Find the exact values of q when p = 20. ii) Find dp dq and dp dq2 and hence show that the equation dp P- = 0 dq has no...
-
Solve 5 tan 20 -8 (0 < < 360)

Study smarter with the SolutionInn App


