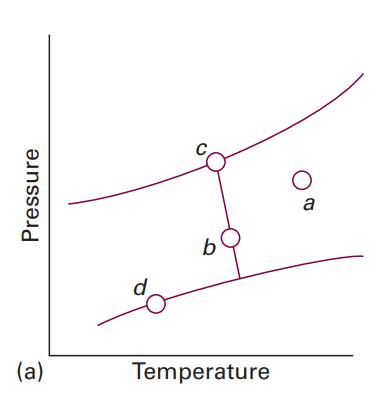
How many phases are present at each of the points marked in Fig. 4.1a? Data in Fig.
Question:
How many phases are present at each of the points marked in Fig. 4.1a?
Data in Fig. 4.1a?
Transcribed Image Text:
Pressure (a) d b Temperature O a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
There are 3 phases present at any point marked in Fig 41a i...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Mr. Lee, clinic director claims that through new procedures he has reduced patient waiting time from present 55 minutes. Before implementing his procedures nationwide, the clinic owner, Ms. Groetken,...
-
Each of the points listed next represents an internal control that may be implemented within a companys accounting information system to reduce various risks. For each point, identify the appropriate...
-
Consider the following phase diagram. What phases are present at points A through H? Identify the triple point, normal boiling point, normal freezing point, and critical point. Which phase is denser,...
-
Carey Company is borrowing $200,000 for one year at 12 percent from Second Intrastate Bank. The bank requires a 20 percent compensating balance. What is the effective rate of interest? What would the...
-
How do firms record prior- period adjustments? .
-
The welded assembly is formed from thin sheet metal having a mass of 19 kg/m 2 . Determine the mass moments of inertia for the assembly about the x- and y-axes. 150 mm 240 mm 80 mm
-
Zachary and Carrie Sexton (the Buyers) were searching for a home in the Kings wood neighborhood of Atlanta, Georgia. The Buyers real estate agent learned that Russell and Linda Sewell (the Sellers)...
-
As sales manager, Joe Batista was given the following static budget report for selling expenses in the Clothing Department of Soria Company for the month of October. As a result of this budget...
-
According to the Civil Code of the Philippines, Article 1 7 6 7 defines partnership as: By the contract of partnership two or more persons bind themselves to contribute money, property, or industry...
-
An average of 15 aircraft accidents occur each year (The World Almanac and Book of Facts, 2004). a. Compute the mean number of aircraft accidents per month. b. Compute the probability of no accidents...
-
The protein lysozyme unfolds at a transition temperature of 75.5 C and the standard enthalpy of transition is 509 kJmol 1 . Calculate the entropy of unfolding of lysozyme at 25.0 C, given that the...
-
Calculate the change in entropy of the system when 10.0 g of ice at 10.0 C is converted into water vapour at 115.0 C and at a constant pressure of 1bar. The constant-pressure molar heat capacity of H...
-
Describe the various forms of economic integration in Europe. What is Brexit, and what are the implications for Great Britains relationship with Europe.
-
Silver Water Motorworks produces small electric motors in lots of 200 with an average yield of 92%. Fifty per cent (50%) of the sub-standard output from the first run through, can be reworked. Direct...
-
Jaeger, Inc. bonds have a 6 . 4 5 % coupon rate with semiannual coupon payments. They have 1 6 years to maturity and a par value of $ 1 , 0 0 0 . What is the current yield of the bond if the bond is...
-
If I want to buy a $500,000 and can get a 30-year mortgage with an APR = 3.5%, what would my monthly payments be? If I can get a 15 year mortgage for 2.5%, how much would I save? I've checked with my...
-
Yuhao paid $220 for a used washing machine and dryer on Kijiji. He paid $85 to a moving company to pick up and deliver washer and dryer. These two events caused nominal GDP to increase by ?
-
Medical Lessors, Inc (MLI) buys an x-ray machine for $200K. On the same day (EOY 0), the firm leases the machine to Radiology, Inc. (RI) for six years, via a six-year, uniform-payment, annuity-due...
-
For the system of Problem P10.20, design a phase lag network to yield the desired specifications, with the exception that a bandwidth equal to or greater than 2 rad/s will be acceptable.
-
Smthe Co. makes furniture. The following data are taken from its production plans for the year. Required: 1. Determine the hazardous waste disposal cost per unit for chairs and for tables if costs...
-
Hexane and perfluorohexane show partial miscibility below 22.70C. The critical concentration at the upper critical temperature is x = 0.355, where x is the mole fraction of C 6 F 14 . At 22.0C the...
-
Some polymers can form liquid crystal mesophases with unusual physical properties. For example, liquid crystalline Kevlar (3) is strong enough to be the material of choice for bulletproof vests and...
-
Refer to the information in Exercise 6.15(b) and sketch the cooling curves for liquid mixtures in which x(B 2 H 6 ) is (a) 0.10, (b) 0.30, (c) 0.50, (d) 0.80, and (e) 0.95. Data in Exercise 6.15(b)...
-
Locating the Next Red Lobster Restaurant From its first Red Lobster in 1968, the chain has grown to 675 locations, with over $1.6 billion in U.S. sales annually. The casual dining market may be...
-
It had been a dream come true for Ash Briggs, a struggling artist living in the San Fran- cisco Bay Area. He had made a trip to the corner grocery store late one Friday after- noon to buy some milk,...
-
Whats the difference between immigration law and regular law practice ? write a detail note on it ?

Study smarter with the SolutionInn App


