Question: The Clapeyron equation does not apply to second-order phase transitions, but there are two analogous equations, the Ehrenfest equations, that do. They are: Where
The Clapeyron equation does not apply to second-order phase transitions, but there are two analogous equations, the Ehrenfest equations, that do. They are:
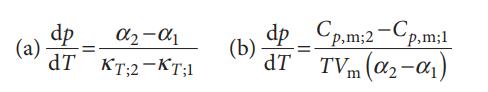
Where α is the expansion coefficient, κT the isothermal compressibility, and the subscripts 1 and 2 refer to two different phases. Derive these two equations. Why does the Clapeyron equation not apply to second-order transitions?
-1 TKT;2-KT;1 dp (a) = dp_Cp,m;2-Cp,m;1 (b) dp= dT TVm (0-01)
Step by Step Solution
3.33 Rating (159 Votes )
There are 3 Steps involved in it
Answer The Ehrenfest equations can be derived by taking the partial derivatives of the Gibbs free en... View full answer

Get step-by-step solutions from verified subject matter experts


