The composition of a liquid phase reaction 2 AB was followed by a spectrophotometric method with the
Question:
The composition of a liquid phase reaction 2 A→B was followed by a spectrophotometric method with the following results:
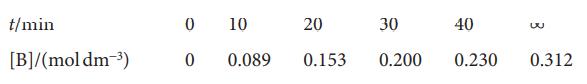
Determine the order of the reaction and its rate constant.
Transcribed Image Text:
t/min [B]/(mol dm-³) 0 0 10 0.089 20 30 0.153 0.200 40 0.230 w 0.312
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Answer Since the order of the reaction will be depending on the concentration of reactions ...View the full answer

Answered By

Marvine mabiya
i am a graduate of kirinyaga university degree of statistics
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Explain how the composition of a small community banks investment portfolio differs, in general, from the composition of a large banks portfolio. Why might mutual funds be attractive to banks?
-
Explain what is involved in determining the composition of a compound of C, H, and O by combustion.
-
Why would breast feeding affect the composition of a baby's intestinal microbiota?
-
Using monthly data for Meta since its IPO date of 31/12/2019, estimate equity beta for Meta. Compare your estimate of Metas equity beta with Facebooks beta provided by Yahoo Finance and comment on...
-
Employees of a state correctional facility were required to attend a seventy-five minute training program entitled Gays and Lesbians in the Workplace. The program was designed to show the facilitys...
-
Five 0.25 m 2 quadrats were placed randomly in an area of grassland in the UK. The percentage of each quadrat occupied by each species of plant was estimated to the nearest 5% and recorded in the...
-
Heritage, a general contractor, had filed a breach of contract action against an electrical subcontracting firm that had withdrawn its bid right before the contract was to be performed. A jury had...
-
A company manufactures three products: A, B, and C. The company currently has an order for three units of product A, 7 units of product B, and 4 units of product C. There is no inventory for any of...
-
An upstart phone company has only two potential large customers, Firm A and Firm B. Firm A's monthly demand for phone calls is Q1 = 2,800 200p (with p measured in cents) and Firm 2's is Q2 =5,000...
-
Demand for Stuffin Such stuffed animal shells and stuffing/finishing kits manufactured by RoseMarie Limited is increasing, and management requests assistance from you in determining the best sales...
-
The oxidation of HSO 3 by O 2 in aqueous solution is a reaction of importance to the processes of acid rain formation and flue gas desulfurization. R.E. Connick et al. (Inorg. Chem. 34, 4543 (1995))...
-
Conventional equilibrium considerations do not apply when a reaction is being driven by light absorption. Thus the steady-state concentration of products and reactants might differ significantly from...
-
Nestl's financial statements are presented in Appendix B. Financial statements of Petra Foods are presented in Appendix C. Instructions (a) Based on the information in these financial statements,...
-
CONTRACT ELEMENTS: Linda and Greg are planning to get married on April 7, 2022. They have decided to book their wedding reception and banquets at the No-Tell hotel. They have tentatively booked a...
-
A stock has the following dividend schedule each year: dividends of $130 are paid at the end of first, second, and third quarters, while the end of year dividend is $200. Find the price of this stock...
-
Purpose Utilizing one of the identified Social Work Theories; evaluate that theory and how it relates to why individuals in the Hispanic community commit suicide. Instructions Find one to two...
-
Alaska Airlines employed Tellis in Seattle as a maintenance mechanic. His wife was having difficulties with her pregnancy, so on the morning of July 4, 2000, he told his supervisor he needed to take...
-
Thanh has a capital gain from the sale of a business of $1.5 million. His current superannuation balance is $500,000. He has the full CGT cap available. After seeking advice from Thanh's accountant,...
-
Use Exercise 10.7.6 to prove that if f is continuous and periodic on [0. 2Ï] and for k = 0,1,..., then f(x) = 0 for all x [a, b]. 2T f (x) cos(kx) dx = f(x) sin(kx) dx = 0
-
we have to compute the letter grades for a course. The data is a collection of student records stored in a file. Each record consists of a name(up to 20 characters), ID (8 characters), the scores of...
-
The rotational constant for 14 N 2 determined from microwave spectroscopy is 1.99824 cm 1 . Calculate the bond length in 14 N 2 to the maximum number of significant figures consistent with this...
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
Calculate the zero point energies for 1 H 19 F and 2 D 19 F. Compare the difference in the zero point energies to k B T at 298 K.
-
write the definite interval mite integral for the Riemann sum: lim 3 4 3 4 4 4 21 [(5+2) + (5+2+2)+(5+2+3)+(5+2)] n n n - n 3 n
-
Can you solve this problem, 1 4 [ [ 0 4x (x + y) 16 y dy dx
-
Why might one prefer to use the logarithmic differentiation for finding derivatives of functions that have another function as an exponent?

Study smarter with the SolutionInn App


