The following data have been obtained for the adsorption of H2 on the surface of 1.00 g
Question:
The following data have been obtained for the adsorption of H2 on the surface of 1.00 g of copper at 0°C. The volume of H2 below is the volume that the gas would occupy at STP (0°C and 1 atm).
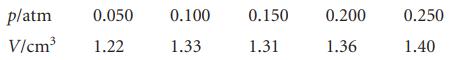
Determine the volume of H2 necessary to form a monolayer and estimate the surface area of the copper sample. The density of liquid hydrogen is 0.708 g cm−3.
Transcribed Image Text:
p/atm 0.050 V/cm³ 1.22 0.100 1.33 0.150 1.31 0.200 1.36 0.250 1.40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Solution The gas adsorption isotherm is given by In the above equ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following data have been obtained for the adsorption of H 2 on the surface of 1.00 g of copper at 0 C. The volume of H2 below is the volume that the gas would occupy at STP (0 C and 1 atm)....
-
The following data have been obtained for the liquid-vapour equilibrium compositions of mixtures of nitrogen and oxygen at 100 kPa. T/K 77.3 78 80 82 84 86 88 90.2 X (O2) 0 10 34 54 70 82 92 100...
-
Following data have been obtained for machining AA390 Aluminum, a Si-Al alloy. Compute the K and n values for the Taylor tool life equation. How do these n values compare to the typical values?...
-
Please help me calculate the current assets and current liabilities. Cash and cash equivalents Deposits Marketable securities Inventory Property & equipment, net Loan to shareholders Notes receivable...
-
The manager of the Cottonwood Grille recently selected a random sample of 18 customers and kept track of how long the customers were required to wait from the time they arrived at the restaurant...
-
(EPS with Options, Various Situations) Zambrano Companys net income for 2010 is $40,000. The only potentially dilutive securities outstanding were 1,000 options issued during 2009, each exercisable...
-
The distributed loading is supported by the three suspender bars. AB and EF are made of aluminum and CD is made of steel. If each bar has a cross-sectional area of 450 mm 2 , determine the maximum...
-
Refer to Problem 6-64. Develop a net material requirements plan assuming that there are currently 150 units of Part A, 40 units of Part B, 50 units of Subassembly C, and 100 units of Part F currently...
-
Critically analyze the following question. PROVIDE REFERENCES What job factors are most important in the workplace of today?
-
For the fourbar linkage shown, P is located at the midpoint of the coupler. 1. Find the orientation of the crank and rocker (theta2 and theta4) at the two toggle positions. Note that these are also...
-
The enthalpy of adsorption of CO on a surface is found to be 120 kJ mol 1 . Estimate the mean lifetime of a CO molecule on the surface at 400 K.
-
The volume of oxygen gas at 0C and 101 kPa adsorbed on the surface of 1.00 g of a sample of silica at 0C was 0.284 cm 3 at 142.4 Torr and 1.430 cm 3 at 760 Torr. What is the value of V mon ?
-
In problem, for the given functions and , find: (a) (f ° g)(4) (b) (g ° f)(2) (c) (f ° f)(1) (d) (g ° g)(0) 3 f(x) 8(x) = V x + 1'
-
What is the purpose and benefit of the polymerase chain reaction (PCR)?
-
Suppose an accountant uncovers evidence of financial fraud within their organization, but reporting it may lead to severe consequences for their colleagues and the company. What ethical principles...
-
4. Consider the function. f(x, y, z)=x+y+z+sin (az) - e-(2+y +2). Write a code that evaulates the Hessian matrix for any choice of r, y and 2. 5. Generate a random vector in matrix with entries = [1,...
-
If the points shown as squares in the plot of volume versus temperature shown above had been included in the calculation of the trendline, would the calculated value of C be too high, too low, or...
-
Premier Bank and Trust is considering giving Waterway Company a loan. Before doing so, they decide that further discussions with Waterway's accountant may be desirable. One area of particular concern...
-
Draw the resonance forms of the half-chair conformation of the oxonium ion transition state of the lysozyme reaction.
-
What is an access control list?
-
Which of the following systems are isolated? a) A bottle of wine b) A tightly sealed, perfectly insulated thermos bottle c) A tube of toothpaste d) our solar system. Explain your answers.
-
Why do the z and y components of the velocity not change in the collision depicted in Figure 1.2? Figure 1.2 mvx mvx
-
A mixture of 2.10 10 3 g of O 2 , 3.88 10 -3 mol of N 2 , and 5.25 10 20 molecules of CO are placed into a vessel of volume 5.25 L at 12.5C. a. Calculate the total pressure in the vessel. b....
-
Performance Products Corporation makes two products, titanium Rims and Posts. Data regarding the two products follow: Direct Labor- Hours per unit Rims Posts 0.70 0.70 Annual Production 26,000 units...
-
How well does this document supports users in taking action or making decisions, as well as how well the document accomplishes inferred purposes of whoever created the document? How this document...
-
Describe/Define the "Innovation Sweet Spot" and identify the three (3) components that intersect to achieve it The ISS is the merging of the 3 driving forces of design innovation Feasible, Viable,...

Study smarter with the SolutionInn App


