The mean activity coefficients for aqueous solutions of NaCl at 25C are given below. Confirm that they
Question:
The mean activity coefficients for aqueous solutions of NaCl at 25°C are given below. Confirm that they support the Debye–Hückel limiting law and that an improved fit is obtained with the extended law.
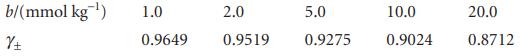
Transcribed Image Text:
b/(mmol kg ¹) 土 1.0 0.9649 2.0 0.9519 5.0 0.9275 10.0 0.9024 20.0 0.8712
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Solution Here we know k A 12ln 12k A 05885 ln 1 04021 ln 2 Since the DebyeHckel law is v...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The mean activity coefficients for aqueous solutions of NaCl at 25 C are given opposite. Confirm that they support the DebyeHckel limiting law and that an improved fit is obtained with the Davies...
-
The volume of an aqueous solution of NaCl at 25 C was measured at a series of molalities b, and it was found that the volume fitted the expression v=1003+16.62x+1.77x 3/2 +0.12x 2 where v=V/cm 3 , V...
-
The mean activity coefficients of KCI in three dilute aqueous solutions at 25C are 0.927 (at 5.0 mmol kg-I), 0.902 (at 10.0 mmol kg-I), and 0.816 (at 50.0 mmol kg"). Estimate the value of B in the...
-
Zolezzi Incorporated is preparing its cash budget for March. The budgeted beginning cash balance is $29,000, Budgeted cash receipts total $100,000 and budgeted cash disbursements total $91,000. The...
-
Why does a dealer offer to trade only a fixed amount at the bid and ask prices?
-
Steady-state temperatures at selected nodal points of the symmetrical section of a flow channel are known to be T2 = 95.47C, T3 = 117.3C, T5 = 79.79C, T6 = 77.29C, T8 = 87.28C, and T10 = 77.65C. The...
-
A convergent-divergent nozzle has an exit throat area ratio of 3.0. It is to be supplied with air. Find: (a) The first, second, and third critical pressure ratios; (b) The exit plane Mach number in...
-
On January 1, 2015, Eddy Corporation had retained earnings of $650,000. During the year, Eddy had the following selected transactions. 1. Declared cash dividends $120,000. 2. Corrected overstatement...
-
Question 2 a) Draw an annotated sketch showing the main features of the oedometer apparatus. What is this apparatus used for? b) Explain the term "isochrone of excess pore pressure". [6 marks] [4...
-
Determine the equivalent annual worth for years 1 through 10 of a uniform series of payments of $20,000 that begins in year 3 and ends in year 10. Use an interest rate of 10% per year. Also, write...
-
The enthalpy of fusion of anthracene is 28.8 kJ mol 1 and its melting point is 217C. Calculate its ideal solubility in benzene at 25C.
-
The osmotic pressure of solutions of polystyrene in toluene were measured at 25C and the pressure was expressed in terms of the height of the solvent of density 1.004 g cm 3 : Calculate the molar...
-
Before preparing financial statements for the current year, the chief accountant for Patel Ltd. provided the following information regarding the accounting for dividends and stock splits: 1. Patel...
-
You only have 500 words or so to work with in the HW assignment (Roche case study) and you can summarize the article a little bit to provide context. The bulk of the case study report should focus on...
-
Define Organizational Culture, describe the key functions of Value Chain, and explain the four line functions that includes Human Resource Management.
-
Two shafts, each of 65 mm diameter, are connected by the gears shown. The shaft at A is fixed and G is 77 GPa. Knowing that a torque T = 1000 N-m is applied at D. determine (a) the maximum shearing...
-
Is there any resource that you could consider as a distinctive competence for Yunnan Lucky Air?
-
1. Briefly describe how a composite growth rate of demand for hotel rooms can be calculated. 2. In estimating total sales revenue for a coffee shop in a proposed new hotel, why is it important to...
-
Given a fixed set of players N, each coalition T N determines a unanimity game uT (example 1.48) defined by 1. For each coalition S c N, recursively define the marginal value of a coalition by...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
As a variation of the preceding problem explore the consequences of increasing the energy separation of the B and C orbitals (use S=0 for this stage of the calculation). Are you justified in ignoring...
-
Write down the secular determinants for (i) anthracene (1), (ii) phenanthrene (2) within the Hckel approximation and using the C2p orbitals as the basis set. 1 Anthracene 2 Phenanthrene
-
What is the physical significance of the Coulomb and resonance integrals?
-
what ways does the concept of biocultural diversity broaden our understanding of the relationship between human societies and biodiversity, and how can this concept inform more holistic conservation...
-
Examine the Year-to-Year Total Return Data in the table below Compute the annual Risk Premiums for Stocks and Bonds Compute the average returns for stocks, bonds, and bills. Compute the standard...
-
Compute common-size percents for the following comparative Income statements spanning 2 years. Comparative Income Statements For Years Ended December 31 Current Yeal Sales Cost of goods sold Gross...

Study smarter with the SolutionInn App


