The osmotic pressure of solutions of polystyrene in toluene were measured at 25C and the pressure was
Question:
The osmotic pressure of solutions of polystyrene in toluene were measured at 25°C and the pressure was expressed in terms of the height of the solvent of density 1.004 g cm−3:
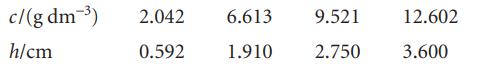
Calculate the molar mass of the polymer.
Transcribed Image Text:
c/(g dm-³) h/cm 2.042 6.613 0.592 1.910 9.521 2.750 12.602 3.600
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Assuming molar mass x then since CRT wh...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 298 K, the osmotic pressure of a glucose solution is 10.50 atm. Calculate the freezing point of the solution. The density of the solution is 1.16 g/mL.
-
The concentration dependence of the osmotic pressure of solutions of a macromolecule at 20C was found to be as follows: Determine the molar mass of the macromolecule and the osmotic virial...
-
The osmotic pressure of 0.010 M solutions of CaCl2 and urea at 25C are 0.605 atm and 0.245 atm, respectively. Calculate the van't Hoff factor for the CaCl2 solution.
-
A geostationary satellite orbiting at a height 36,000km operates at 4GHz using a transmitter antenna that has a gain of 15dB. On earth the receiver antenna has a gain of 45dB. Calculate; (a) Free...
-
The SEC regulates American stock markets. However, NYSE members have committees that carry out a host of self-regulatory activities. NYSE members are profit seekingwhy would they self-regulate...
-
The steady-state temperatures (oC) associated with selected nodal points of a two-dimensional system having a thermal conductivity of 1.5 W/m K are shown on the accompanying grid. (a) Determine the...
-
Air is flowing in the converging-diverging nozzle shown in Fig. P11.52. Determine the three critical pressure ratios and the Mach numbers immediately upstream and immediately downstream from the...
-
Eddings Company had a beginning inventory of 400 units of Product XNA at a cost of $8.00 per unit. During the year, purchases were: Eddings Company uses a periodic inventory system. Sales totaled...
-
1. What is the entry mode to a foreign country that Starbucks developed in their international strategy? Why choose this entry mode? Please explain in detail. 2. Has Starbucks adopted a...
-
Derive the state table and state diagram of the sequential circuit shown in Fig. 2: Ck Fig. 2
-
The mean activity coefficients for aqueous solutions of NaCl at 25C are given below. Confirm that they support the DebyeHckel limiting law and that an improved fit is obtained with the extended law....
-
A water carbonating plant is available for use in the home and operates by providing carbon dioxide at 5.0 atm. Estimate the molar concentration of the soda water it produces.
-
Of the types of radioactive decay studied in this chapter, which is least likely to damage you upon external exposure? Which is most likely? Explain fully.
-
How do you ensure that our organization's human resource planning aligns with its long-term strategic goals?"
-
1) Describe any two of the following terms concepts: Expectancy theory of motivation. Maslow's need hierarchy. Recognition need. 2)Define any two of the following terms : Autocratic...
-
Your Capsim Strategy - Low Tech or High Tech? Will you manage your company as a cost leader or differentiator? Will you orient your company to a broad or a niche customer base? Based on your answers...
-
By making use of relevant examples, describe the four areas in which business logistics strategy, tactics and operations can enhance the long - term wealth of a business?
-
A) Assuming that there are only 2 airline companies in the U.S. flying between Newark and Miami, Delta and Continental, what is the ((Nash) Equilibrium) or price that each company in the following...
-
What is the linear hull of the vectors {(1, 0), (0,2)} in R2?
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
In a particular photoelectron spectrum using 21.21 eV photons, electrons were ejected with kinetic energies of 11.01 eV, 8.23 eV, and 5.22 eV. Sketch the molecular orbital energy level diagram for...
-
Evaluate the bond order of each Period 2 homonuclear diatomic molecule.
-
Set up the secular determinants for the homologous series consisting of ethene, butadiene, hexatriene, and octatetraene and diagonalize them by using mathematical software. Use your results to show...
-
Gather than focus on the treatment of chronic disease, policies that influence population health tend to emphasize prevention and wellness; the reduction or elimination of waste, and the eradication...
-
A corporation is an organization created by law and treated as a legal entity, literally a legal person, that is separate from and independent of the individuals who are involved in it. As a legal...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...

Study smarter with the SolutionInn App


