The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are
Question:
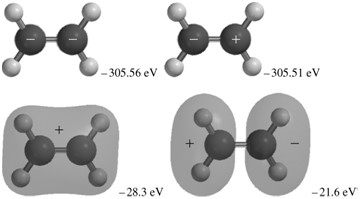
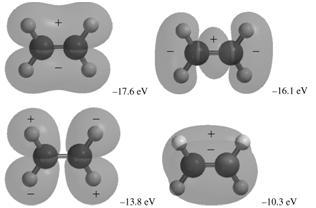
Transcribed Image Text:
+. -305.51 eV -305.56 eV -28.3 eV -21.6 eV -17.6 eV -16.1 ev -13.8 eV -10.3 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The MOs corresponding to the energies 30356 eV and 30351eV are the inphase and outofphase combinatio...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
83+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of hydrogen cyanide are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as...
-
Indicate the type of solute-solvent interaction (Section 11.2) that should be most important in each of the following solutions: (a) CCl4 in benzene (C6H6), (b) Methanol (CH3OH) in water, (c) KBr in...
-
Juanita Torres is 35-years old and lately has been working with her financial planner. She is attempting develop a long-term savings and investment program. She has been thinking about all the major...
-
When f (t) = sin t and g (t) = cos t, is h (t) = f (t) g (t) even, odd, or neither? Explain.
-
A ship constructed of \(2.50 \times 10^{6} \mathrm{~kg}\) of steel is roughly shaped like a box of length \(50.0 \mathrm{~m}\), width \(20.0 \mathrm{~m}\), and height \(20.0 \mathrm{~m}\). (a) What...
-
Discuss major concerns in controlling and reducing the spread of hospital-acquired infections.
-
Abridged balance sheets and income statements along with relevant note disclosures from Revlon Inc.'s 2011 annual report are presented below. Required: a. Compute the following key financial ratios...
-
Rearrange the code blocks below to build a proper elif statement. Drag from here if my_var 5: elif my_var
-
Make a circuit which gives the absolute value of a 4-bit binary number. Use four full adders. four multiplexers. and four inverters. Assume negative numbers are represented in 2's complement. Recall...
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Use the geometrical construction shown in Example Problem 24.8 to derive the electron MO levels for cyclobutadiene. What is the total energy of the molecule? How many unpaired electrons will the...
-
What are the five major leadership styles identified on the Leadership Grid ?
-
Explain the factors in the external environment that have led to the increased interest in strategic reward management;
-
Outline the strengths and limitations for diversity management of the equal opportunities approach.
-
What do you consider to be the strengths and limitations of the equal opportunities and the managing diversity approaches to diversity management?
-
The Tibbett and Britten Group (TBG) is a UK-based international logistics service provider. Its main customers are retailers and manufacturers, to whom it provides warehouse, distribution and...
-
For federal income tax purposes a partnership may be composed of: a. Two or more individuals b. An individual and a corporation c. A trust, an estate, and an individual d. Two or more partnerships e....
-
For 5% compounded continuously In problem, find the effective rate of interest.
-
Compare and contrast debt financing and equity financing as ways of starting a new business. Does one have an overall advantage over the other? What situation is more favorable to the use of debt...
-
At 518C, the half-life for the decomposition of a sample of gaseous acetaldehyde (ethanal) initially at 363 Torr was 410 s. When the pressure was 169 Torr, the half-life was 880 s. Determine the...
-
The second-order rate constant for the reaction is 0.11 dm 3 mol 1 s 1 . What is the concentration of ester after (a) 10 s, (b) 10 min when ethyl acetate is added to sodium hydroxide so that the...
-
Sucrose is readily hydrolysed to glucose and fructose in acidic solution. The hydrolysis is often monitored by measuring the angle of rotation of plane polarized light passing through the solution....
-
Janice will need to pay $200 at the end of every month for the next 12 months, except for the payment of the 8th month. What is the present value, assuming a rate of 4%, compounded quarterly?
-
List and explain two management tools in the planning process and two measurable performance indicators. Explain in detail.
-
What does a high PE tell us about the value of the stock price (over or under valued)? What does a low PE tell us about the value of the stock price (over or under valued)? Be specific with your...

Study smarter with the SolutionInn App


