The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are
Question:
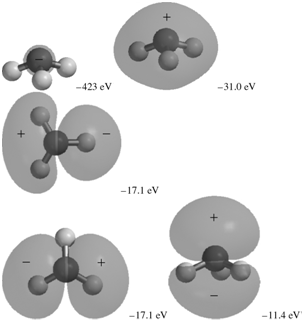
Transcribed Image Text:
-31.0 eV -423 eV -17.1 ev -17.1 eV -11.4 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The MO with an energy of 423 eV corresponds to the 1s AO on N It is a loca...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of hydrogen cyanide are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as...
-
Indicate the type of solute-solvent interaction (Section 11.2) that should be most important in each of the following solutions: (a) CCl4 in benzene (C6H6), (b) Methanol (CH3OH) in water, (c) KBr in...
-
What is the is the effect of abolition of an import quota on (i) national saving, (ii) domestic investment, (iii) NCO, (iv) the real exchange rate, and (v) net exports?
-
(a) Use a graphing utility to complete the table. (b) Make a conjecture about the relationship between sin and sin(180 ). 0 20 0 40 60 80 sin e sin(180 0)
-
In an air duct running along the outside of a building, air flowing through the duct has a mass density of \(1.20 \mathrm{~kg} / \mathrm{m}^{3}\) and is moving at \(5.38 \mathrm{~m} / \mathrm{s}\)....
-
If a nurse knows that a piece of equipment is defective, should the nurse attempt to fix the problem? Discuss your answer.
-
As of December 31, 2015, its first year in business, Kukui Company had taxable temporary differences totaling $110,000. Of this total, $45,000 relates to current items. Kukui also had deductible...
-
float f; int i; scanf("%f %i", &f, &i); What will be the values stored in f and i after scanf statement if following values are entered 12.5 1 12 45 12 23.2 12.1 10 12 1 Write a program to verify...
-
1. Compare the first-digit frequency in the transactions in this table below with Benfords Law. What are the results? 2. Could fraud be occurring in thisorganization? AMOUNT DESCRIPTION CHECK NO...
-
Use the VSEPR method to predict the structures of the following: a. PF 3 b. CO 2 c. BrF 5 d. SO 2 3
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
College tuition has risen at a pace faster than inflation for more than two decades, according to an article in USA Today . The following data indicate the average college tuition (in 2003 dollars)...
-
In which of the following situations will the divorced custodial parent be entitled to the dependency exemption for the child? a. The noncustodial parent provides \(\$ 1,500\) of support for the...
-
Use the information in the table from problem 4 to draw the marginal cost curve and the average total cost curve for the first 5 loaves made by the Island Bakery. If you were to continue these curves...
-
LB Corporation purchased a business car in June 2018 for \(\$ 62,000\). The car weighs 4,000 pounds and will be used 100 percent of the time in the business. Compute the largest depreciation...
-
Assume the business equipment purchased in March was instead purchased in November. Based on these facts, compute the MACRS depreciation for the business equipment, car, and office furniture for...
-
Identify the various formats for organising the employment relationship at a range of levels.
-
After the Challenger disaster in 1986, a study was made of the 23 launches that preceded the fatal flight. A mathematical model was developed involving the relationship between the Fahrenheit...
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
The reaction mechanism involves an intermediate A. Deduce the rate law for the reaction. AA+A (fast) A+B P (slow)
-
A reaction 2 A P has a second-order rate law with k = 3.50 10 4 dm 3 mol 1 s 1 . Calculate the time required for the concentration of A to change from 0.260 mol dm 3 to 0.011 mol dm 3 .
-
Consider the dimerization 2 A A 2 , with forward rate constant k a and backward rate constant k b . (a) Derive the following expression for the relaxation time in terms of the total concentration of...
-
S&J Catering is a small catering business operating in Western Sydney. Business partners, Jack and Simon, established the business a year ago. To start up the business, the partners contributed...
-
In 20X1, Lee was hurt in the course of his employment with Foster Farms. In 20X1, Lee was paid the following amounts from Foster Farms' worker's compensation insurance company: $30,000 for a physical...
-
Briefly describe tax administrative matters including: Tax collection and withholding mechanisms (PAYG withholding, PAYG instalments).

Study smarter with the SolutionInn App


