What do you expect the electronic spectrum to look like for the ground and excited states shown
Question:
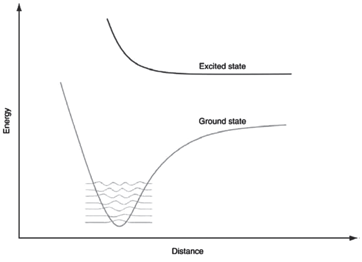
Transcribed Image Text:
Excited state Ground state Distance ABsoug
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The electronic spectrum will have no ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Do you expect the viscosity of glycerol, C3H5(OH)3, to be larger or smaller than that of 1-propanol, C3H7OH? Explain? (a) Glycerol (b) 1-Propanol
-
Consider the tetrahedral anions VO43- (orthovanadate ion), GrO42- (chromate ion), and MnO4 (permanganate ion). (a) These anions are isoelectronic. What does this statement mean? (b)Would you expect...
-
Do you expect the light given off by (a) a neon sign or (b) an incandescent lightbulb to be continuous in distribution or in the form of a line spectrum? Explain.
-
A manufacturing company reports the following information for the month of May. Note: Assume all raw materials were used as direct materials. Activities for May Advertising expense Raw materials...
-
In Exercises 1-4, find the exact values of the six trigonometric functions of the angle . 1. 2. 3. 4. 8. 13
-
The concentration of mercury in a lake has been monitored for a number of years. Measurements taken on a weekly basis yielded an average of 1.20 mg/m3 (milligrams per cubic meter) with a standard...
-
Retro Furniture Ltd suffered a severe drop in sales and profit performance for the year ended 30 June 2025. The income statement revealed that net sales were \($1\) 500 000 with a profit of \($310\)...
-
Review the February 26 and March 25 transactions for Success Systems (SP 5) from Chapter 5. Required 1. Assume that Lyn Addie is an unmarried employee. Her $ 1,000 of wages are subject to no...
-
5. Find a theta notation for the following: T(n) = 4T(n/2) + n Explain how you got your solution. 6. Given the following code: fun (n) { if (n == 1) return; else { for x = 1 to n { x = x + 1; print...
-
As soon as she graduated from college, Kay began planning for her retirement. Her plans were to deposit $500 semiannually into an IRA (a retirement fund) beginning six months after graduation and...
-
Because internal conversion is in general very fast, the absorption and fluorescence spectra are shifted in frequency as shown in Figure 25.10. This shift is crucial in making fluorescence...
-
Why are the spectra of the individual molecules in the bottom trace of Figure 25.15 shifted in frequency? 10,000 Molecules x1 1,000 x10 x100 100 10 Frequency Absorption
-
Draw a simple process flow diagram illustrating the different steps a guest goes through to receive one specific service, such as embarkation, arranging shore excursions, etc.
-
During the period 19602007, earnings of the S&P 500 Index companies have increased at an average rate of 8.18 percent per year, and the dividends paid have increased at an average rate of 5.9 percent...
-
Orpheum Productions in Nevada is considering three mutually exclusive alternatives for lighting enhancements to one of its recording studios. Each enhancement will increase revenues by attracting...
-
Record the following details relating to a carpet retailer for the month of November 2017 and extract a trial balance as at 30 November 2017. 2017 Nov 1 Started in business with 15,000 in the bank. 3...
-
Extranuclear inheritance occurs due to a. chromosomes that have become detached from the spindle apparatus during meiosis. b. genetic material that is found in chloroplasts and mitochondria. c....
-
What is the output of the following? A. false 1 B. false 2 C. true 1 D. true 2 E. Does not compile 5: var line = new String("-"); 6: var anotherLine = line.concat(""); 7: System.out.print(line ==...
-
The sum of any six consecutive Fibonacci numbers is always divisible by 4. Select any six consecutive Fibonacci numbers and show that for your selection this statement is true.
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
For how long on average would an H atom remain on a surface at 298 K if its desorption activation energy were (a) 15 kJ mol 1 , (b) 150 kJ mol 1 ? Take 0 = 0.10 ps. For how long on average would the...
-
The adsorption of a gas is described by the Langmuir isotherm with K = 0.85 kPa 1 at 25C. Calculate the pressure at which the fractional surface coverage is (a) 0.15, (b) 0.95.
-
(a) Discuss the main structural features of the electrical double layer. (b) Distinguish between the electrical double layer and the Nernst diffusion layer.
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


