Question: Because internal conversion is in general very fast, the absorption and fluorescence spectra are shifted in frequency as shown in Figure 25.10. This shift is
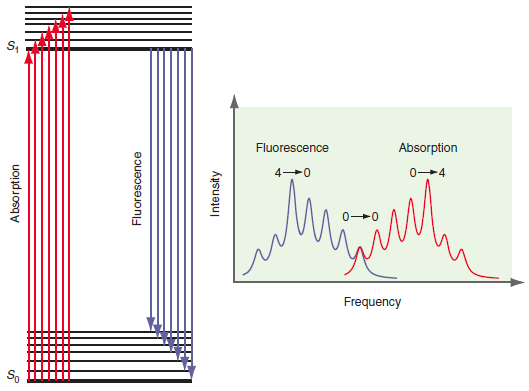
Fluorescence Absorption 0-4 Frequency So Absorption Fluorescence Intensity
Step by Step Solution
3.37 Rating (166 Votes )
There are 3 Steps involved in it
In the experiment the sample is constantly being illuminated with light to effect the transition of ... View full answer

Get step-by-step solutions from verified subject matter experts


