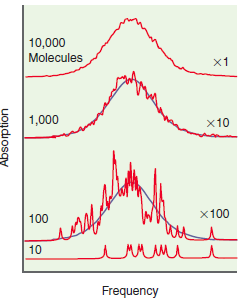
Why are the spectra of the individual molecules in the bottom trace of Figure 25.15 shifted in
Question:
Transcribed Image Text:
10,000 Molecules x1 1,000 x10 x100 100 10 Frequency Absorption
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
In a solution different molecules do not all ...View the full answer

Answered By

Surojit Das
I have vast knowledge in the field of Mathematics, Business Management and Marketing. Besides, I have been teaching on the topics Management leadership, Business Administration, Human Resource Management, Business Communication, Accounting, Auditing, Organizer Behaviours, Business Writing, Essay Writing, Copy Writing, Blog Writing since 2020. It is my personality to act quickly in any emergency situations when students need my services. I am very professional and serious in every questions students asked me at the time of dealing any projects. I have been serving detailed, quality, properly analysed research paper through the years.
4.80+
91+ Reviews
278+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Does it make sense to distinguish individual NaCl molecules in a salt crystal? What about individual H 2 O molecules in an ice crystal? Explain.
-
The following spectra are taken from a compound that is an important starting material for organic synthesis. Determine the structure, first by considering each spectrum individually, then by...
-
Why is a type O individual considered a universal blood donor? Why is a type AB individual considered a universal acceptor?
-
For Questions consider the S-N curve provided below for this same material and situation. Stress & (MPa) 400 300 B. 175 MPa C. 200 MPa 200 100 10 C. 350 MPa D. 400 MPa E. It will never fail P-0.99...
-
In Exercises 1-4, use trigonometric identities to transform the left side of the equation into the right side (0 < < / 2). 1. tan cot = 1 2. cos sec = 1 3. tan cos = sin 4. cot sin = cos
-
David Johnson has decided to start a business and is a new client of your small public accounting practice called Trent Accounting Services. He has come to seek your advice about what form of...
-
Describe the differences between an express and an implied contract.
-
Consider a blending tank that has the same dimensions and nominal flow rates as the storage tank in Exercise 2.13 but incorporates a valve on the outflow line that is used to establish flow rate w3....
-
1 . Explain Engels Law 2. How can Engels law be used to explain the increase in demand for workers in the service industries?
-
An oil company purchased an option on land in Alaska. Preliminary geologic studies assigned the following prior probabilities. P(high-quality oil) = 0.50 P(medium-quality oil) = 0.20 P(no oil) = 0.30...
-
What do you expect the electronic spectrum to look like for the ground and excited states shown in the figure below? Excited state Ground state Distance ABsoug
-
Suppose you obtain the UV photoelectron spectrum shown here for a gas-phase molecule. Each of the groups corresponds to a cation produced by ejecting an electron from a different MO. What can you...
-
100 mL of water at 15C is placed in the freezer compartment of a refrigerator with a coefficient of performance of 4.0. How much heat energy is exhausted into the room as the water is changed to ice...
-
Assess the complexity of issues associated with aligning culture to an organisations strategic direction;
-
Generally, what is "at risk"?
-
The municipality of Viewpoint, Kansas, wishes to attract local businesses. As part of a development program, it donated a building with a basis of \(\$ 50,000\) and a value of \(\$ 200,000\) plus...
-
Evaluate the importance of the cultural perspective on strategic international human resource management.
-
What are the benefits of installment reporting?
-
In problem, find the domain of the composite function f g.
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
The average time for which an oxygen atom remains adsorbed to a tungsten surface is 0.36 s at 2548 K and 3.49 s at 2362 K. Find the activation energy for desorption. What is the pre-exponential...
-
The enthalpy of adsorption of CO on a surface is found to be 120 kJ mol 1 . Estimate the mean lifetime of a CO molecule on the surface at 400 K.
-
The following data have been obtained for the adsorption of H2 on the surface of 1.00 g of copper at 0C. The volume of H 2 below is the volume that the gas would occupy at STP (0C and 1 atm)....
-
5. Recursive method; what is printed if we call mystery (6, 12); public static void mystery (int x, int y) { if (x > y) { System.out.print ("/"); else if (x = y) { } else { System.out.print ("=" + y...
-
Simmonds Products has spent $158,500 (sunk cost) on research to develop low-fat imitation wine. The firm is planning to spend $210,000 on a machine to produce new wine. Shipping cost of $60,000 and...
-
In particular, you were introduced to three decision-making techniques -- majority rule, minority rule by authority or expertise, and consensus rule -- each with their own pros and cons. Using these...

Study smarter with the SolutionInn App


