Question: What would the intensity versus frequency plot in Figure 25.10 look like if fluorescence were fast with respect to internal conversion? Fluorescence Absorption 0-4 Frequency
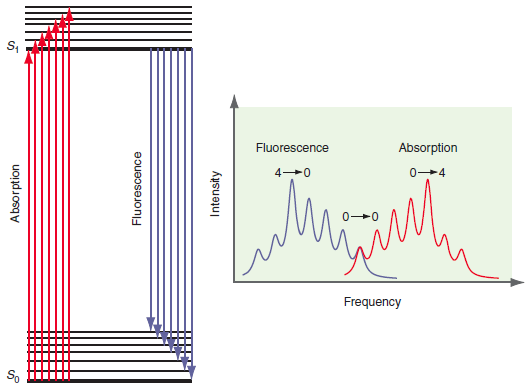
Fluorescence Absorption 0-4 Frequency So Absorption Fluorescence Intensity
Step by Step Solution
3.39 Rating (183 Votes )
There are 3 Steps involved in it
If fluorescence were fast with respe... View full answer

Get step-by-step solutions from verified subject matter experts


