Where would Kb emission peaks for Ti, Se, and Zr be found in Figure 21-31? Why are
Question:
Where would Kb emission peaks for Ti, Se, and Zr be found in Figure 21-31? Why are they not labeled?
In Figure 21-31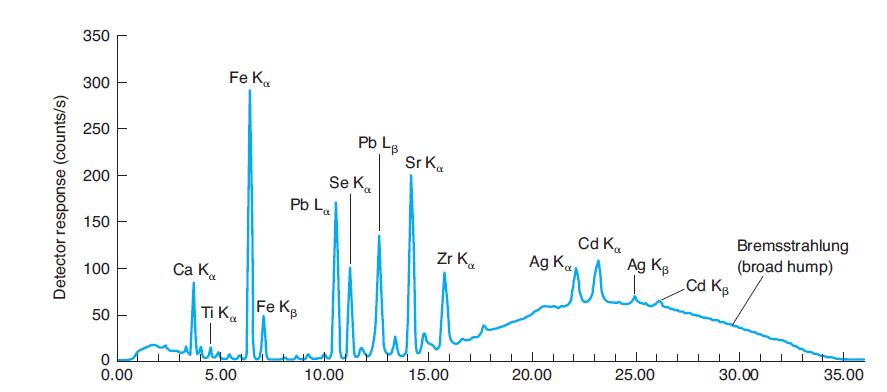
Transcribed Image Text:
350 Fe K. 300 250 Pb L. Sr K. Se K. Pb La 200 150 Cd K. Ag K Bremsstrahlung (broad hump) Cd Kg Zr K. Ag Kg 100 Ca K. Ti Ka Fe KB 50 25.00 30.00 35.00 5.00 10.00 15.00 20.00 0.00 Detector response (counts/s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
For Ti Kbeta line is at 493181kev so next to the ka...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is a hedge fund? Why are they not a recommended investment for most investors?
-
Why are higher resolution monochromators found in ICP atomic emission spectrometers than in flame atomic absorption spectrometers?
-
Why are La and Lb peaks, but not Ka and Kb peaks for lead identified in Figure 21-31? Why are La and Lb peaks not identified for iron in Figure 21-31? In Figure 21-31 350 Fe K. 300 250 Pb L. Sr K. Se...
-
Medallion and RIEF (Renaissance Institutional Equity Fund) are both managed by Renaissance Technologies. How do they differ in terms of asset classes, dollar capacity, average holding period of each...
-
What is an organization's responsibility in regard to sexual harassment among coworkers or supervisor subordinate pairs? Do you think that Daryl Kolendich responded appropriately to the problem?
-
Simplify each and express answers using positive exponents only. If rational exponents appear in final answers, convert to radical form. (A) 93/2 (D) (2x-3/41/4) 4 (B) (-27)4/3 8x1/21/3 (E) x2/3 (C)...
-
You lean out of your dorm window, which is \(12 \mathrm{~m}\) above the ground, and toss a \(0.12 \mathrm{~kg}\) ball up to a friend at a window \(11 \mathrm{~m}\) above you. (a) What is the slowest...
-
In her will, Teressa granted a life estate to Amos in certain real estate, with remainder to Brenda and Clive in joint tenancy. All the residue of Teressas estate was left to Hillman College. While...
-
6. Five Star reality estimates its WACC to be 10 percent. It is considering investments in the following independent projects for the next year. Property Rate of Return Risk (a) 8th Street 14% High...
-
This is the second year that your firm is auditing JJ Company, which is developing a new drug for a rare form of cancer. The company is controlled by Jack, who purchased the shares from the previous...
-
Explain why X-ray fluorescence is observed when matter absorbs X-rays of sufficient energy. Why does each element have a unique X-ray signature?
-
How much energy in kJ/mol is released when nitrogen emits Ka radiation at 0.392 keV? Compare the Ka energy to 945 kJ/mol, which is the energy required to break the triple bond in N 2 (one of the...
-
A woman who used to drink about one liter of regular cola every day switches to diet cola (zero calorie) and starts eating two slices of apple pie every day. Is she now consuming fewer or more...
-
the demand for tylenol, a chemical sold in a perfectly competitive market, is given by Q = 200 - 2P, where the quantity is measured in pounds. the private marginal cost is MC = 20 + 0.75Q, but...
-
What is the value of x after the following statements? int x, y, z; y = 10; z = 3; x = y + z = 3; -
-
Target has posted a job description for the following open position: Responsibilities of the job include collating, preparing and interpreting reports, budgets, accounts, commentaries, and financial...
-
For a recent year, McDonald's (MCD) company-owned restaurants had the following sales and expenses (in millions): Sales Food and paper Payroll and employee benefits Occupancy and other expenses...
-
What two areas in the Cash Flow report can you determine when a client starts to take supplemental withdrawals from their portfolio?
-
In an Earth laboratory, an astronaut measures the length of a rod to be 1.00 m. The astronaut takes the rod aboard a spaceship and flies away from Earth at speed 0.5c. Is the length of the rod...
-
List four items of financial information you consider to be important to a manager of a business that has been operating for a year.
-
The diprotic acid H2A has pK1 = 4.00 and pK2 = 8.00. (a) At what pH is [H2A] = [HA-]? (b) At what pH is [HA-] = [A2-]? (c) Which is the principal species at pH 2.00: H2A, HA-, or A2-? (d) Which is...
-
The base B has pKb = 5.00. (a) What is the value of pKa for the acid BH+? (b) At what pH is [BH+] = [B]? (c) Which is the principal species at pH 7.00: B or BH+? (d) What is the quotient [B]/[BH+] at...
-
The acid HA has pKa = 4.00. Use Equations 9-17 and 9-18 to find the fraction in the form HA and the fraction in the form A- at pH = 5.00. Does your answer agree with what you expect for the quotient...
-
Choose the correct answer- a. The Basel I framework increases the quality of capital and increases the coverage of risks. b. The Basel III framework increases the quality of capital and increases the...
-
Question 2 Heat Inc. sells computer hard drives, and the market is competitive (i.e., sales price will not be affected by Heat's sales volume). Its EBIT for 2022 was $69,000 when the sales quantity...
-
brfm17h_ch10.04m 1.Only Visited, Not Judged 2.Only Visited, Not Judged 3.Not Answered 4.Not Answered 5.Not Answered 6.Not Answered 7.Not Answered 8.Not Answered 9.Not Answered 10.Not Answered...

Study smarter with the SolutionInn App


