Why are La and Lb peaks, but not Ka and Kb peaks for lead identified in Figure
Question:
Why are La and Lb peaks, but not Ka and Kb peaks for lead identified in Figure 21-31? Why are La and Lb peaks not identified for iron in Figure 21-31?
In Figure 21-31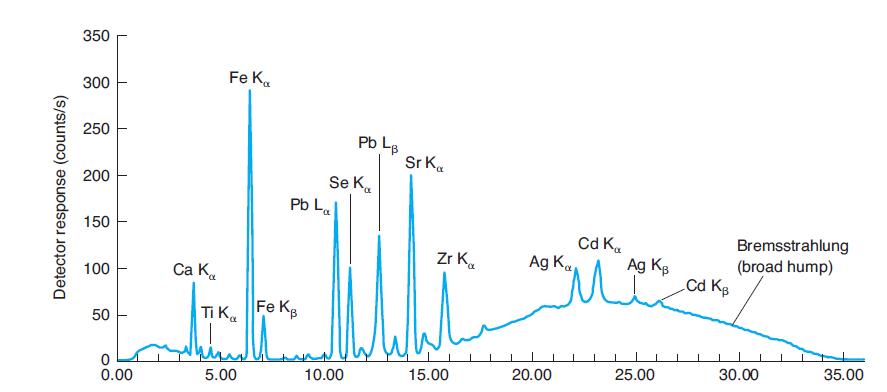
Transcribed Image Text:
350 Fe K. 300 250 Pb L. Sr K. Se K. Pb La 200 150 Cd K. Ag K Bremsstrahlung (broad hump) Cd Kg Zr K. Ag Kg 100 Ca K. Ti Ka Fe KB 50 25.00 30.00 35.00 5.00 10.00 15.00 20.00 0.00 Detector response (counts/s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Answer There are four fundamental processes that will p...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why will a block of iron float in mercury but sink in water?
-
Why do maximizing EPS and maximizing value not necessarily lead to the same conclusion about the optimal capital structure?
-
Why are the multiplet splittings in Figure 28.9 not dependent on the static magnetic field? J12 J42 (yB/2m)(01- 2) Frequency Intensity -----
-
Wollongong Group Ltd, of New South Wales, Australia, acquired its factory building about 10 years ago. For several years the company has rented out a small annex attached to the rear of the building....
-
If you were Erin Dempsey, what would you do?
-
Simplify, and express the answers using positive exponents only. (A) (2x 3 ) (3x 5 ) (B) x 5 x 9 (C) x 5 /x 7 (D) x 3 /y 4 (E) (u 3 v 2 ) 2 (F) (G) -2
-
A \(30-\mathrm{kg}\) child jumps to the ground from the top of a fence that is \(2.0 \mathrm{~m}\) high. You analyze the problem using upward as the positive \(x\) direction. (a) Taking \(x=0\) to be...
-
Tupper Memorial Hospital received from a donor a $50,000 contribution and a $50,000 pledge payable in one year. The donor required that the funds be used for heart research. Explain how these...
-
You are the prime minister of a new African nation that is oil rich. Your currency is the Zambia (made up name). Would you choose to let your currency float, be pegged or do something else? Provide...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
Where would Kb emission peaks for Ti, Se, and Zr be found in Figure 21-31? Why are they not labeled? In Figure 21-31 350 Fe K. 300 250 Pb L. Sr K. Se K. Pb La 200 150 Cd K. Ag K Bremsstrahlung (broad...
-
How much energy in kJ/mol is released when nitrogen emits Ka radiation at 0.392 keV? Compare the Ka energy to 945 kJ/mol, which is the energy required to break the triple bond in N 2 (one of the...
-
In Exercises 1-4, approximate the logarithm using the properties of logarithms, given logb 2 0.3562, logb 3 0.5646, and logb 5 0.8271. 1. logb 10 2. logb 2/3 3. logb 0.04 4. logb 2
-
A company that makes soccer balls for professional soccer teams wants to understand the amount of rework in a given manufacturing process. The process has three steps with the following yields: 99%,...
-
Janet's employer provides her a company car. in exchange, janet pays a predetermined amount for personal use of the car. under the commuting value method, what is the least amount that janet must pay?
-
What will be the force of electric repulsion between two small spheres placed 1.0 m apart, if each has a deficit of 108 electrons?
-
Nuclear power plants provide 64.6 % of the total of 5723 MW of electrical energy consumed during a hot summer's day in a certain province. How many megawatts (MW) of electrical energy did the nuclear...
-
What is not an indication a client may have a credit problem? Explain please.
-
Harry and Sally are on opposite sides of the room at a wedding reception. They simultaneously (in the frame of the room) take flash pictures of the bride and groom cutting the cake in the center of...
-
Match each of the key terms with the definition that best fits it. _______________ A record of the sequence of data entries and the date of those entries. Here are the key terms from the chapter. The...
-
A dibasic compound, B, has pKb1 = 4.00 and pKb2 = 6.00. Find the fraction in the form BH2 2+ at pH 7.00, using Equation 9 - 19. Note that K1 and K2 in Equation 9-19 are acid dissociation constants...
-
Write the chemical reactions whose equilibrium constants are Kb1 and Kb2 for the amino acid proline. Find the values of Kb1 and Kb2.
-
What fraction of ethane-1,2-dithiol is in each form (H2A, HA-, A2-) at pH 8.00? at pH 10.00?
-
A. Let's start with a simple calculation to set the stage for our analysis. Suppose you wish to estimate the value of Heinz by means of a perpetuity (i.e., assume that the free cash flows of the last...
-
Case 2 12-shelf unit. 150 inches/shelf. 21,000 inches of current records. 6,000 projected inches needed for future. Calculate the number of filing units to be purchased. Show all calculations.
-
Which condition is necessary for a transaction to be considered a hostile takeover? Group of answer choices The target firm must be publicly traded. The target firm must be privately held. The...

Study smarter with the SolutionInn App


