Mass spectral interpretation. The compound C9H4N2Cl6 C 9 H 4 N 2 Cl 6 is a by-product
Question:
Mass spectral interpretation. The compound C9H4N2Cl6 C9H4N2Cl6 is a by-product found in chlorinated pesticides.
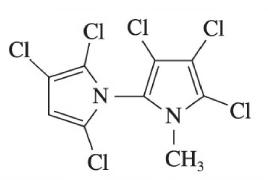
a. Verify that the formula for rings + double bonds agrees with the structure.
b. Find the nominal mass of C9H4N2Cl6.C9H4N2Cl6.
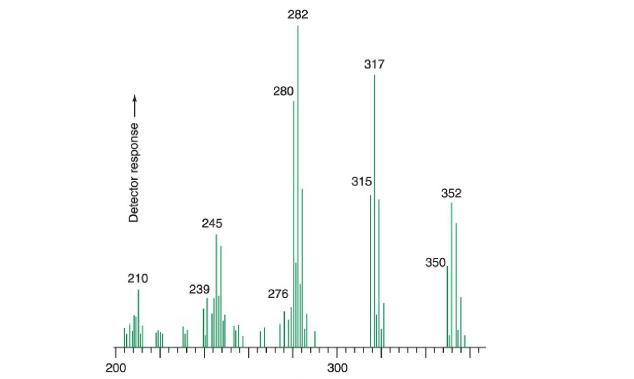
c. Suggest an assignment for m/z350,m/z350, 315,315, 280,280, 245245, and 210210 in the high-mass region of the electron ionization mass spectrum.
Transcribed Image Text:
CI Cl Cl N- CI CI N I CH3 Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a The formula for rings double bonds for the compound C9H4N2Cl6 is C9H4N...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Verify that the formula for t of Theorem 14.4 can be written as Theorem 14.4 Under the assumptions of normal regression analysis, Is a value of a random variable having the t distribution with n 2...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
Suggest a composition for the halogen compound whose mass spectrum is shown in the figure. Assign each of the major peaks. 100 127 173 160 25 158 162 7 48 HT 79 91 160 170 208 35 160 173 IITI'T1 30...
-
Consider the following graph. 2 7 6 C 10 10 H 11 E 12 15 14 (a). Find a minimum spanning tree of the graph using Kruskal's algorithm. List the edges in the order they are put into the tree. (b)....
-
For the data presented in Table 11.9, construct a confidence ellipse for β0 and β1 with confidence coefficient 0.95. Table 11.9 Data for Exercise 1 87427 00077 12100 67890...
-
On November 1, 20X1, Gerakos Corporation sold software and a six-month technical support contract to a customer for $80,000. Gerakos sells the same software without technical support for $60,000. It...
-
True or False: If \(E R R>M A R R\), then \(I R R>E R R>M A R R\).
-
A job cost sheet of Chamberlin Company is given below. Instructions (a) Answer the following questions. (1) What are the source documents for direct materials, direct labor, and manufacturing...
-
(1 point) A local polka band wants to make and sell CDs of its songs. Suppose it costs $ 1000 to record, S 450 to edit, and S 350 for album artwork, and suppose each CD that is manufactured will cost...
-
Posey, a member, provides bookkeeping services for Giant Construction, a sole owner company of ten employees. On a monthly basis the owner sends Posey copies of check stubs and bank statements so...
-
Nanoparticles with a gold core and organothiol outer shell with well-defined stoichiometries Aux(SR)yAu x (SR) y i can be prepared and isolated. a. The MALDI-time-of-flight mass spectrum of the...
-
Calculate the theoretical masses of the sucralose species in Figure 22-14 at nominal masses of 395,395, 397,397, 399,399 and 401.401. Find the difference in ppm between observed and calculated...
-
Making hiring decisions is one of the most importantand difficultdecisions a manager makes. Without good information, managers have almost no chance of making the right choice. The process begins...
-
The program will introduce the use of if-statements, and plan out the steps for it We need to have a program that will let the user enter a base value and an exponent value. They will pick from a...
-
Mary is single mother with 12 years old daughter. She is the only person supporting her family. Her mother, Cathy 60 years old lives with her, has no income. Cathy is disabled and has T2201 approved....
-
What appraisal technique lists traits (i.e. communication, teamwork, and responsibility) and a range of performance values with points (i.e. satisfactory, unsatisfactory, average) for each of the...
-
Find a story or article from newspaper that is related to some aspect of intercultural communication, such as problems encountered in the process of Acculturation. Write a report discussing those...
-
What makes a great leader? Management theorist Simon Sinek suggests, it's someone who makes their employees feel secure, who draws staffers into a circle of trust. But creating trust and safety...
-
Steam at 3 MPa and 400oC enters an adiabatic nozzle steadily with a velocity of 40 m/s and leaves at 2.5 MPa and 300 m/s. Determine (a) The exit temperature and (b) The ratio of the inlet to exit...
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
Detection limit. Low concentrations of Ni2+ -EDTA near the detection limit gave the following counts in a mass spectral measurement: 175, 104, 164, 193, 131, 189, 155, 133, 151, 176. Ten measurements...
-
What are the three parts of quality assurance? What questions are asked in each part and what actions are taken in each part?
-
Detection limit. A sensitive chromatographic method was developed to measure sub-part per-billion levels of the disinfectant by-products iodate (IO 3 ), chlorite (ClO 2 ), and bromate (BrO 3 ) in...
-
In order to provide good care for a culturally diverse patient population, it is important to gain some understanding of their ways of being (their belief systems, their traditions, their feelings...
-
Describe how men and women experience divorce differently -. use the following headings for this response -"Men Experience Divorce" and "Women Experience Divorce". Explain why the divorce rate rose...
-
1. Authentic leaders do not fake their leadership. Support your response with evidence from the readings and videos. 2. Authentic leaders lead from conviction. Support your response with evidence...

Study smarter with the SolutionInn App


