Use Figure 25-33 for the following questions: a. What pHpH would be best for the separation of
Question:
Use Figure 25-33 for the following questions:
a. What pHpH would be best for the separation of benzoic acid, 4-4-nitrophenol, and 3-3-methylbenzoic acid?
b. What pHpH would be best for the separation of benzoic acid, 3-3-methylbenzoic acid, and 4-4-methylaniline?
c. What pHpH would be best for separation of 4-4-nitrophenol, 4-4-methylaniline, and codeine on a typical C18-silica C18-silica column?
Figure 25-33
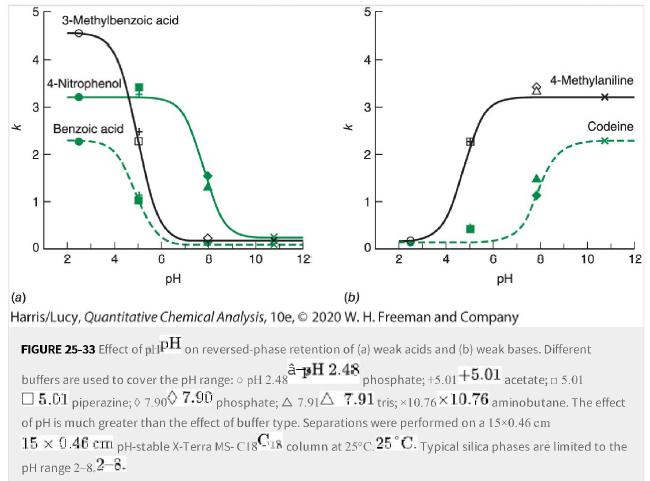
Transcribed Image Text:
K 3 2 1 3-Methylbenzoic acid 4-Nitrophenol Benzoic acid 2 G 0 10 12 pH K 5 4 3 2 1 0 I 2 4-Methylaniline Codeine 10 12 pH (b) (a) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company FIGURE 25-33 Effect of plPH on reversed-phase retention of (a) weak acids and (b) weak bases. Different à pH 2.48 buffers are used to cover the pH range: o pH 2.48 phosphate; +5.01+5.01 acetate; n 5.01 5.01 piperazine; 7.900 7.90 phosphate; A 7.91A 7.91 tris; 10.76×10.76 aminobutane. The effect of pH is much greater than the effect of buffer type. Separations were performed on a 15x0.46 cm 15 x 0.46 cm pH-stable X-Terra MS-C1818 column at 25°C 25 °C. Typical silica phases are limited to the pH range 2-8.2-8.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Answer a The best pH for the separation of benzoic acid 44nitroph...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Refer to below graph Figure 141 to answer the following questions a What is the Federal Fund Rate at the initial level of reserves supply b How does the Fed hit its federal funds rate target of 2
-
Select all the correct option for the following questions 1. Money a. Is more efficient than barter. b. Makes trades easier. c. Allows greater specialization. d. All of the above are correct. 2....
-
Highlight the correct option for the following questions 31. When a firm expands in the production process to supply its own raw materials and intermediate products or to provide its own sales or...
-
Hybrid Corporation began operations in 2018 and during that year purchased equity investments. It owns less than 20% of the voting shares for all companies whose stock it purchased. The year-end cost...
-
Show that the M.L.E. of 2 in the general linear model is given by Eq. (11.5.7).
-
Consider the following three complexes: (Complex 1) [Co(NH 3 ) 5 SCN] 2+ (Complex 2) [Co(NH 3 ) 3 Cl3] 2+ (Complex 3) CoClBr 5NH 3 Which of the three complexes can have (a) Geometric isomers (b)...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
a. Assume that Caro-Life wants to maximize the average sales potential of its ten offices. Where should it locate offices and what is the population and geographic area associated with each office?...
-
Who developed the relational model, when, and why?
-
Drilling Company uses activity-based costing and provides this information: Drilling has just completed 80 units of a component for a customer. Each unit required 100 parts and 3 machine hours. The...
-
Simulating a separation with a spreadsheet. Use the spreadsheet in Figure 25-39 to simulate the chromatograms for =0.75= 0.75 and =0.56= 0.56 in Figure 25-40. Figure 25-39 Figure 25-40 A 1...
-
The figure shows reversed-phase retention data for three compounds. a. Identify whether compounds A, B, and C are weak acids or bases. For each compound, what is the pKap K a and the retention factor...
-
You are in the last stages of evaluating laptop vendors for a major hardware upgrade and standardization project for your firm. You will be purchasing a total of 1,200 new laptops to deploy to the...
-
Case Study: Coke vs. Pepsi This case study discussion is about branding. Formulate a response responding to the specific questions below rather than those within in the case study. Review this case...
-
Applications Utility is generally applied by financial specialists to such develops as the detachment bend, which plot the blend of items that an individual would acknowledge to keep a given degree...
-
6.What will $150,000 grow to be in 15 years if it is invested in an account with a quoted annual interest rate of 10% with monthly compounding of interest? 7.How many years will it take for $200,000...
-
Take on the role of a marketing professional for Goodwill Industries International nonprofit organisation to develop a persuasive marketing strategy for good. Your goal is to help society while being...
-
explain to me while in this code, even though I am able to ask the user for an integer, it is now printing out the name twice. For example, if the user enters john and then enters john for the...
-
Steam enters a steady-flow turbine with a mass flow rate of 13 kg/s at 600°C, 8 MPa, and a negligible velocity. The steam expands in the turbine to a saturated vapor at 300 kPa where 10 percent...
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
What is the true mass of water weighed at 24C in the air if the apparent mass is 1.0346 0.0002 g? The density of air is 0.0012 0.0001g/mL and the density of balance weights is 8.0 0.5 g/mL. The...
-
Round each number as indicated: (a) 1.236 7 to 4 significant figures (b) 1.238 4 to 4 significant figures (c) 0.135 2 to 3 significant figures (d) 2.051 to 2 significant figures (e) 2.005 0 to 3...
-
Twelve dietary iron tablets were analyzed by the gravimetric procedure in Section 1-4 and the final mass of Fe2O3 (FM 159.688) was 0.2774 0.0018 g. Find the average mass of Fe per tablet.(Relative...
-
A (closed) umbrella 1m long is placed 10cm off the optical axis of a convex mirror. It is oriented parallel to the optical axis. The umbrella's tip is 50cm from the mirror (thus the end of its handle...
-
The figure below depicts a planet with an atmosphere comprised of two layers of greenhouse gases. The orange arrow depicts the incoming solar radiation. The different red arrows depict the longwave...
-
A circuit consisting of two batteries, two identical resistors, and some wires is represented by the circuit diagram below. +12V +4V Construct a graph showing the value of the electric potential (V)...

Study smarter with the SolutionInn App


