Place each charge form of alanine under the pH condition where it would be the predominant form.
Question:
Place each charge form of alanine under the pH condition where it would be the predominant form. The pKa values for the carboxyl group and amino group of alanine are approximately 2.3 and 9.7, respectively.
If you answer any part of this question incorrectly. a single red X will appear indicating that one or more of the items were placed incorrectly. 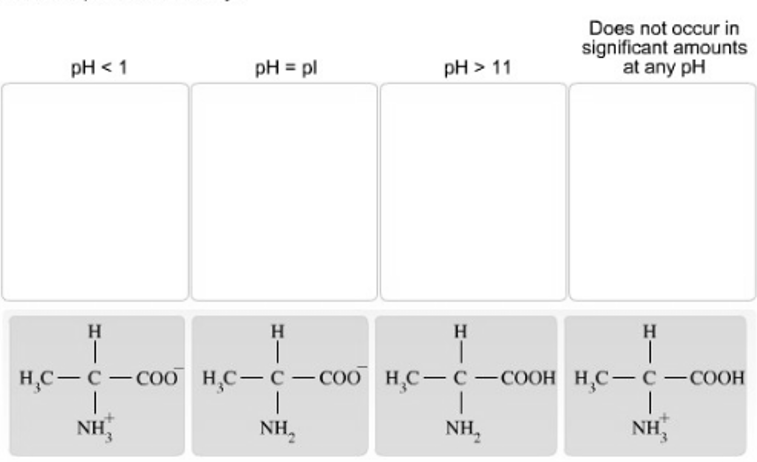 Construct a two-step synthesis of (?) leucine by dragging the appropriate formulas into the bins. Note that each bin will hold only one item. and not all of the given reagents or structures will be used.
Construct a two-step synthesis of (?) leucine by dragging the appropriate formulas into the bins. Note that each bin will hold only one item. and not all of the given reagents or structures will be used.
If one or more statements are incorrectly placed, a single red X will appear on the top left of the left bin. This does not necessarily mean the reagent placed in the left bin is wrong, but that there is a mistake somewhere. 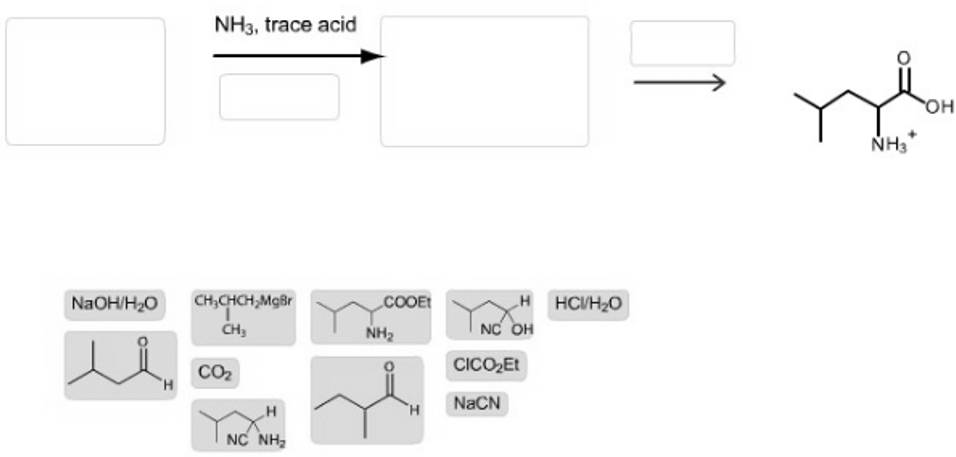

Investment Analysis and Portfolio Management
ISBN: 978-0538482387
10th Edition
Authors: Frank K. Reilly, Keith C. Brown




