Question: A gas mixture contains each of the following gases at the indicated partial pressure. N2 219 torr O2 109 torr He 249 torr What
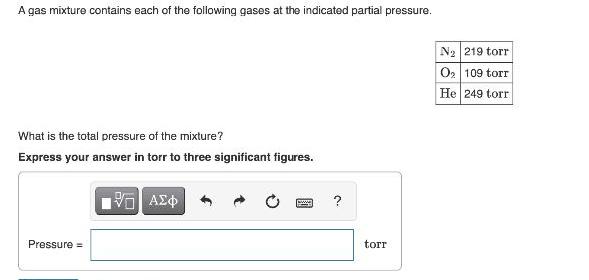
A gas mixture contains each of the following gases at the indicated partial pressure. N2 219 torr O2 109 torr He 249 torr What is the total pressure of the mixture? Express your answer in torr to three significant figures. Pressure = torr
Step by Step Solution
3.39 Rating (161 Votes )
There are 3 Steps involved in it
To find the total pressure ... View full answer

Get step-by-step solutions from verified subject matter experts


