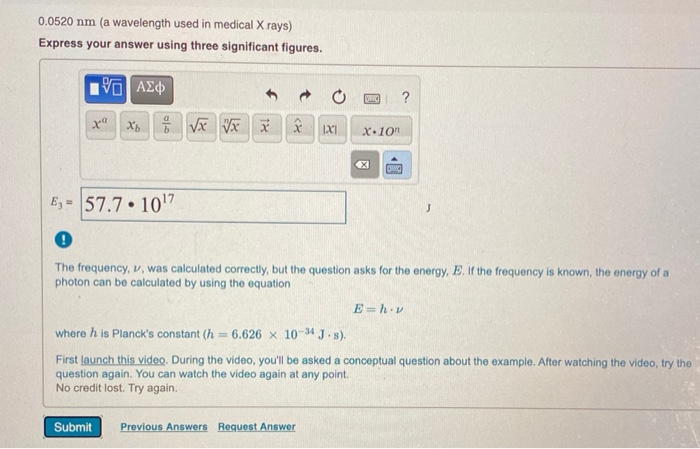
0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 0.0520 nm (a wavelength used in medical X rays) Express your answer using three significant figures. 195] ΑΣΦ xa Xb√x Vxx E₁= 57.7 10¹7 ● XX Submit IXI D Previous Answers Request Answer X.10n ? ! The frequency, v, was calculated correctly, but the question asks for the energy, E. If the frequency is known, the energy of a photon can be calculated by using the equation OMC where h is Planck's constant (h= 6.626 x 10-34 J-s). First launch this video. During the video, you'll be asked a conceptual question about the example. After watching the video, try the question again. You can watch the video again at any point. No credit lost. Try again. E=h.v MISSED THIS? Read Section 8.2 (Pages 312-322); Watch KCV 8.2, IWE 8.2. Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths.
Expert Answer:
Related Book For 

Calculus Early Transcendentals
ISBN: 9781337613927
9th Edition
Authors: James Stewart, Daniel K. Clegg, Saleem Watson, Lothar Redlin
Posted Date:
Students also viewed these chemistry questions
-
If calculate f (46) (3). Express your answer using factorial notation: n! = 1 2 3 (n 1) n x46 + x45 + 2 f(x) 1 + x
-
A gas can be described by the following equation of state: P = RT/ (v-b) where b= 5 E-4 m3/mol and cp(ig)=35 J/molk. (a) Develop an expression for cp(real) for this gas. (b) One mole of this gas...
-
X rays with initial wavelength 0.0665 nm undergo Compton scattering. What is the longest wavelength found in the scattered x rays? At which scattering angle is this wavelength observed?
-
Please answer the following. Show your calculations for potential partial credit. Assume the expected return on the market is 14 percent and the risk-free rate is 4 percent. A.)What is the expected...
-
Is it unethical for a company to provide a sustainability report, but provide no assurance on the reliability of the information contained therein?
-
Regent Corp. is evaluating three competing types of equipment. Costs and cash flow projections for all three are given in the following table. Which would be the best choice based on payback period?...
-
Texas Inpatient Consultants, LLLP, is a partnership that employs physicians to deliver medical care to hospitalized patients of other physicians. Texas Inpatient recruited Julius Tabe, M.D., to work...
-
1. Joshua Vermier of Sacramento, California, received a raise after his first year on the job to $43,800 from his initial salary of $42,000. What was Joshuas raise stated as a percentage? Inflation...
-
A researcher collected self-report data on life satisfaction, social support, and depression. She found the following correlations: (1) life satisfaction and depression, (8) = -.77, p < .01; (2)...
-
Because Natalie has had such a successful first few months, she is considering other opportunities to develop her business. One opportunity is the sale of fine European juicing machines. The owner of...
-
How did doctors create my belly button?
-
Check your own state statute for its version of the UCC.
-
For each of the following studies, decide whether to illustrate the described outcomes with a line graph or a bar graph; then create graphs that accurately portray the outcomes. 1. In a study of the...
-
Which of the following traversals always gives the sorted sequence of the elements in a BST? Preorder Ignored Postorder Undefined
-
Explain the functions of a clearinghouse associated with a futures exchange.
-
How do margin requirements in the futures market differ from margin requirements in the cash market?
-
World Oil Company can buy two types of crude oil: light oil at a cost of $25 per barrel, and heavy oil at $22 per barrel. Each barrel of crude oil, when refined, yields three products: gasoline, jet...
-
What is beacon marketing? What are digital wallets?
-
The deck of a bridge is suspended 275 feet above a river. If a pebble falls off the side of the bridge, the height, in feet, of the pebble above the water surface after t seconds is given by y 275 -...
-
Use logarithmic differentiation to find the derivative of the function. y = 1/
-
Find the limit or show that it does not exist. do q + 6q 4 lim 49? - 39 + 3 9>00
-
What are FMEAs three risk assessment factors?
-
Cignet Plastics Corporation is a contract plastics die-casting house serving a wide range of clients. Over the years, Cignet has been a favored supplier of precision die castings for a major producer...
-
Contrast decision making with problem solving.

Study smarter with the SolutionInn App



