Question: 1. (19 points) An adsorption study was conducted by adding varying amounts of activated carbon to a series of air samples consisting of 1
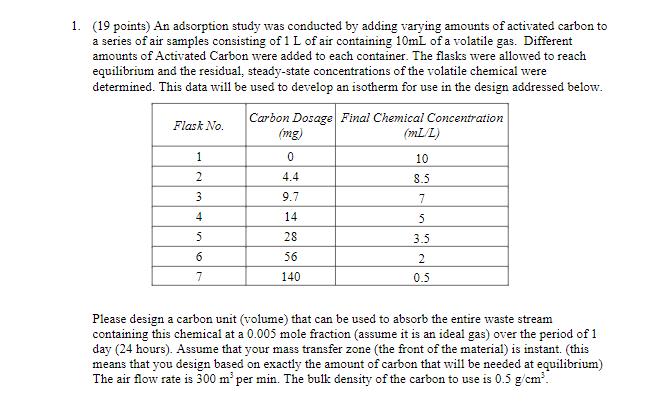
1. (19 points) An adsorption study was conducted by adding varying amounts of activated carbon to a series of air samples consisting of 1 L of air containing 10mL of a volatile gas. Different amounts of Activated Carbon were added to each container. The flasks were allowed to reach equilibrium and the residual, steady-state concentrations of the volatile chemical were determined. This data will be used to develop an isotherm for use in the design addressed below. Carbon Dosage Final Chemical Concentration Flask No. (mg) (mL/L) 1 0 10 2 4.4 8.5 3 9.7 7 4 14 5 5 28 3.5 6 56 2 7 140 0.5 Please design a carbon unit (volume) that can be used to absorb the entire waste stream containing this chemical at a 0.005 mole fraction (assume it is an ideal gas) over the period of 1 day (24 hours). Assume that your mass transfer zone (the front of the material) is instant. (this means that you design based on exactly the amount of carbon that will be needed at equilibrium) The air flow rate is 300 m per min. The bulk density of the carbon to use is 0.5 g/cm.
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


