A ternary mixture of methanol, ethanol, and water is distilled in a sieve-tray column to obtain a
Question:
A ternary mixture of methanol, ethanol, and water is distilled in a sieve-tray column to obtain a distillate with not more than 0.01 mol% water. The feed to the column is as follows: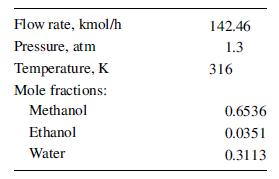
Transcribed Image Text:
Flow rate, kmol/h Pressure, atm Temperature, K Mole fractions: Methanol Ethanol Water 142.46 1.3 316 0.6536 0.0351 0.3113
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The aim is to obtain a distillate with not more than 001 mol water The feed to the column is as foll...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
A ternary mixture of methanol, ethanol, and water is distilled in a sieve-tray column to obtain a distillate with not more than 0.01 mol% water. The feed to the column is as follows: For a distillate...
-
When a mixture of ethanol and water is distilled at ambient pressure, the products are a distillate of ethanol and water of nearazeotrope composition (89.4 mol% ethanol) and a bottoms product of...
-
Five hundred kmol/h of a liquid mixture of light alcohols containing, by moles, 40% methanol (M), 35% ethanol (E), 15% isopropanol (IP), and 10% normal propanol (NP) is distilled in a sequence of two...
-
Listed below are selected items from the cost-of-quality (COQ) report for Watson Products for last month. Category Amount Rework ........... $ 725 Equipment maintenance ...... 1,154 Product testing...
-
High productivity in the service sector has historically been difficult to achieve. Why was this so? What might be changing in this area that will cause service productivity to increase during the...
-
A researcher surveys every tenth house for average family incomes. Identify the sampling technique used, and discuss potential sources of bias (if any). Explain.
-
Water enters a 5 -ft-wide, 1 -ft-deep channel as shown in Fig. P4.66. Across the inlet the water velocity is \(6 \mathrm{ft} / \mathrm{s}\) in the center portion of the channel and \(1 \mathrm{ft} /...
-
Even with all of Marriotts brands they still do not have any hotel properties in the truly budget category of lodging. Thats a category occupied by properties such as Days Inn and Budgetel. Why do...
-
An empty 150g glass bottle is at room temperature, 22 Celcius, if 270g of boiling water is poured into the bottle, what is the temperature of the bottle and the water when they reach thermal...
-
A 0.7812-g sample of impure Al 2 (CO 3 ) 3 decomposed with HCl; the liberated CO 2 was collected on calcium oxide and found to weigh 0.04380 g. Calculate the percentage of aluminum in the sample.
-
It is required to absorb 96% of the benzene from a gas stream with absorption oil in a sieve-tray column at a nominal pressure of 1 atm. The feed conditions are as follows: Tray geometry is as...
-
A bubble-cap tray absorber is designed to absorb 40% of the propane from a rich gas at 4 atm. The specifications for the entering rich gas and absorbent oil are as follows: (a) Determine the number...
-
The graph of a function is given. (a) Use the graph to predict the number of real zeros and the number of nonreal complex zeros. (b) Find these zeros using the quadratic formula. 21 1 3 y=-x-2 X
-
Global Dominion Access is planning to issue preferred stock. The stock sells for \($140;\) however, if new stock is issued, the company would receive only \($115.\) The par value of the stock is...
-
Energias de Portugals common stock is currently selling for \($27.32.\) Dividends paid last year were \($0.75\). Flotation costs on issuing stock will be 12 percent of market price. The dividends and...
-
Antwerp Water Works is selling for \($67.87\) per share and paid a dividend of \($1.38\) last year. The dividend is expected to grow at 5 percent indefinitely. What is the stocks expected rate of...
-
The preferred stock of Brille Power Co. sells for \($35\) and pays 5 percent of the earnings in dividends. The net price of the security after issuance costs is \($30.\) What is the cost of capital...
-
Essilor Internationals common stock pays a dividend of \($0.85.\) It is currently selling for \($35.54.\) If the firms investors require a 12 percent return on their investment from buying Essilor...
-
Evaluate each expression without using a calculator. Then check your work with a calculator. a. 4 - 2 b. (-3) - 1 c. (1/5)-3 d. 491/2 e. 64-1/3 f. (9/16)3/2 g. -70 h. (3)(2)2 i. (0.6-2)-1/2
-
During the year land was revalued and the surplus reported as Revaluation surplus; and an asset costing 80,000, written down to 38,000, was sold for 40,000. Identify the cost of any non-current...
-
A diploid nucleus of Drosophila melanogaster contains about 3.4 10 8 nucleotide pairs. Assume (1) that all nuclear DNA is packaged in nucleosomes and (2) that an average internucleosome linker size...
-
The relationship between the melting T m and GC content can be expressed, in its much simplified form, by the formula T m = 69 + 0.41 (% GC). (a) Calculate the melting temperature of E. coli DNA that...
-
How could it be demonstrated that the mixing of heatkilled Type III pneumococcus with live Type II resulted in a transfer of genetic material from Type III to Type II rather than a restoration of...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
Marcia Stanton is the new manager of the materials storeroom for Taylor Manufacturing. Marcia has been asked to estimate future monthly purchase costs for part #696, used in two of Taylor's products....
-
Miller Company's contribution format income statement for the most recent month is shown below: Sales (37,000 units) Variable expenses Contribution margin Fixed expenses Net operating income...

Study smarter with the SolutionInn App


