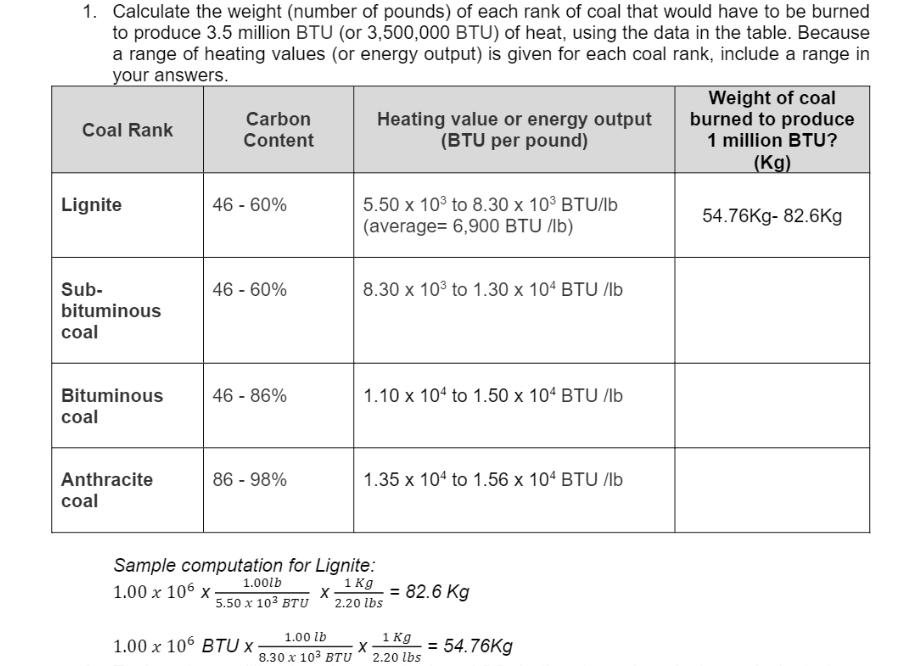
1. Calculate the weight (number of pounds) of each rank of coal that would have to...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
1. Calculate the weight (number of pounds) of each rank of coal that would have to be burned to produce 3.5 million BTU (or 3,500,000 BTU) of heat, using the data in the table. Because a range of heating values (or energy output) is given for each coal rank, include a range in your answers. Coal Rank Lignite Sub- bituminous coal Bituminous coal Anthracite coal Carbon Content 46 - 60% 46 - 60% 46 - 86% 86 - 98% 1.00 x 106 BTU X X 1.00 lb 8.30 x 10³ BTU Heating value or energy output (BTU per pound) 5.50 x 10³ to 8.30 x 10³ BTU/lb (average= 6,900 BTU/lb) 8.30 x 10³ to 1.30 x 104 BTU /lb Sample computation for Lignite: 1.00lb 1.00 x 106 x 1 Kg 2.20 lbs 5.50 x 10³ BTU 1.10 x 104 to 1.50 x 104 BTU /lb 1.35 x 104 to 1.56 x 104 BTU/lb X = 82.6 Kg 1 Kg 2.20 lbs = = 54.76Kg Weight of coal burned to produce 1 million BTU? (Kg) 54.76Kg- 82.6Kg 2. How many billion metric tons of CO₂ have been added to the atmosphere between 1800 and June of 2020? Show all pertinent solutions in the box provided. 3. Compute for the amount of increase in concentration of CO₂ in ppm and in metric tons. Year Atmospheric CO₂ Weight of CO₂ (in billion metric tons) 683.73 852.00 1966 2014 amount of increase from 1966 to 2014 concentration 321 ppm 400 ppm 1. Calculate the weight (number of pounds) of each rank of coal that would have to be burned to produce 3.5 million BTU (or 3,500,000 BTU) of heat, using the data in the table. Because a range of heating values (or energy output) is given for each coal rank, include a range in your answers. Coal Rank Lignite Sub- bituminous coal Bituminous coal Anthracite coal Carbon Content 46 - 60% 46 - 60% 46 - 86% 86 - 98% 1.00 x 106 BTU X X 1.00 lb 8.30 x 10³ BTU Heating value or energy output (BTU per pound) 5.50 x 10³ to 8.30 x 10³ BTU/lb (average= 6,900 BTU/lb) 8.30 x 10³ to 1.30 x 104 BTU /lb Sample computation for Lignite: 1.00lb 1.00 x 106 x 1 Kg 2.20 lbs 5.50 x 10³ BTU 1.10 x 104 to 1.50 x 104 BTU /lb 1.35 x 104 to 1.56 x 104 BTU/lb X = 82.6 Kg 1 Kg 2.20 lbs = = 54.76Kg Weight of coal burned to produce 1 million BTU? (Kg) 54.76Kg- 82.6Kg 2. How many billion metric tons of CO₂ have been added to the atmosphere between 1800 and June of 2020? Show all pertinent solutions in the box provided. 3. Compute for the amount of increase in concentration of CO₂ in ppm and in metric tons. Year Atmospheric CO₂ Weight of CO₂ (in billion metric tons) 683.73 852.00 1966 2014 amount of increase from 1966 to 2014 concentration 321 ppm 400 ppm
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Using the data in the table below, calculate the return for investing in this stock from January 1 to December 31. Prices are after the dividend has been paid (see MyFinanceLab for the data in...
-
Using the data in the table below, complete the table by calculating the cumulative planned and cumulative actual monthly budgets through the end of June. Complete the earned value column on the...
-
Using the data in the table below, estimate the demand function for cod. Prices, dollar per pound Quantity, thousand pounds per day 1.90 1.5 1.35 2.2 1.25 4.4 1.20 5.9 0.95 6.5 0.85 7.0 0.73 8.8...
-
The MRP gross requirements for Item A are shown here for the next 10 weeks. Lead time for A is three weeks and setup cost is $ 10. There is a carrying cost of $ 0.01 per unit per week. Beginning...
-
Determine the break-even contribution margin ratio for a company with a fixed overhead of $92,000 and revenues of $450,000.
-
The closing inventory figure shown on a statement of financial position is always determined by a physical count/computer printout.
-
What is a say-on-pay vote?
-
OfficePlus sells its main product, ergonomic mouse pads, for $12 each. Its variable cost is $5.20 per pad. Fixed costs are $205,000 per month for volumes up to 65,000 pads. Above 65,000 pads, monthly...
-
Question 11pts The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed...
-
Suppose the current USD/euro exchange rate is 1.2000 dollar per euro. The six month forward exchange rate is 1.1950. The six month USD interest rate is 1% per annum continuously compounded? Estimate...
-
Which of the following is the best option to reduce the break-even level of a firm offering a highly elastic commodity? A. Increase variable cost per unit B. Increase sales price per unit 0 C....
-
Two protons, each of mass \(m_{\mathrm{p}}=1.67 \times 10^{-27} \mathrm{~kg}\), collide to produce a particle that has a mass 300 times the mass of each proton. What is the minimum kinetic energy...
-
The mean weight of cows in a population is 520 kg. Animals with a mean weight of 540 kg are used as parents and produce offspring that have a mean weight of 535 kg. What is the narrowsense...
-
Are two consecutive moves \(M_{1}\) and \(M_{2}\) on Rubik's cube commutative; that is, \[M_{1} M_{2}=M_{2} M_{1} ?\]
-
If we assume 10% of the energy put in for crushing/grinding leads to heat generation then will more heat will be generated to break particles from 10 to 5 mm, or from 1 to 0.5 mm?
-
a. Does your state allow covenant marriage? If so, describe its components. b. Explain why you think it is a good or bad idea for every state to force couples to choose between conventional and...
-
A company uses a process costing system. The company uses the weighted-average method and reports the following data for its Welding department. Units Direct materials Percent Complete Conversion...
-
What are the before image (BFIM) and after image (AFIM) of a data item? What is the difference between in-place updating and shadowing, with respect to their handling of BFIM and AFIM?
-
Given the following data: 2ClF(g) + O2(g) Cl2O(g) + F2O(g) H = 167.4 kJ 2ClF3(g) + 2O2(g) Cl2O(g) + 3F2O(g) H = 341.4 kJ 2F2(g) + O2(g) 2F2O(g) H = 43.4 kJ Calculate H for the reaction ClF(g) +...
-
Although we often assume that the heat capacity of a substance is not temperature dependent, this is not strictly true, as shown by the following data for ice: Use these data to calculate graphically...
-
Derive an expression for the relationship between pKa and pKb for a conjugate acidbase pair.
-
Kinney A.. had the following assets and liabilities on the dates indicated. Kinney began business on January 1, 2024, with an investment of 100,000. Instructions From an analysis of the change in...
-
Two items are omitted from each of the following summaries of statement of financial position and income statement data for two proprietorships for the year 2025, Greenes Goods and Solar Enterprises....
-
Listed below are some items found in the financial statements of Min-Seo Promotions. Indicate in which financial statement(s) the following items would appear. a. Service revenue. b. Equipment. c....

Study smarter with the SolutionInn App


