1) Consider the two-stage, adiabatic steam turbine, shown below. Steam at 15 MPa and 600C enters...
Fantastic news! We've Found the answer you've been seeking!
Question:
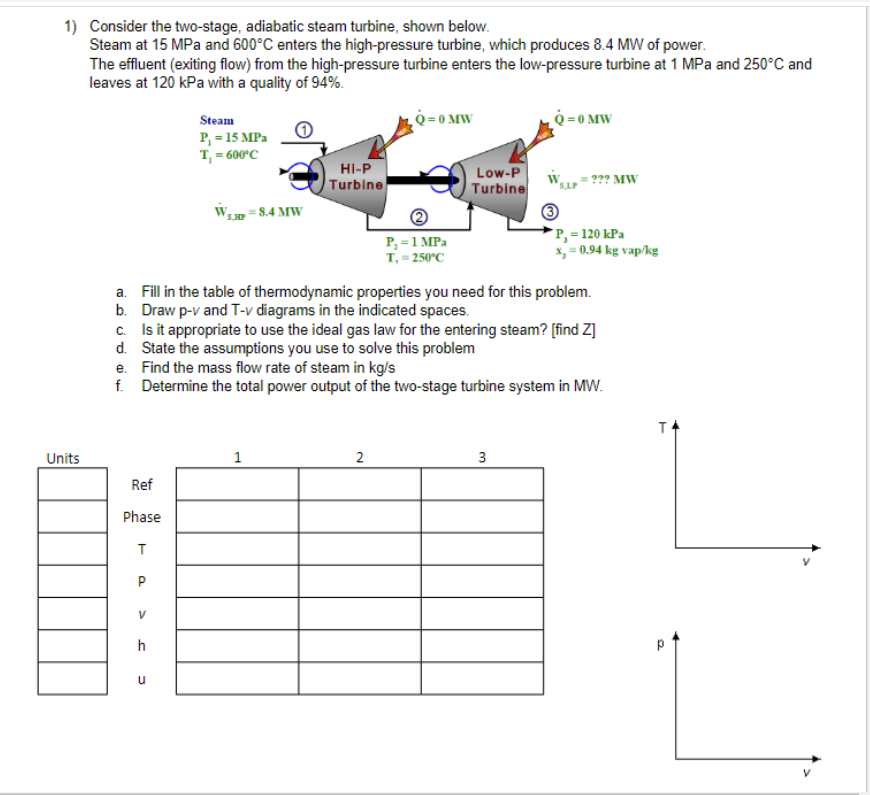
Transcribed Image Text:
1) Consider the two-stage, adiabatic steam turbine, shown below. Steam at 15 MPa and 600C enters the high-pressure turbine, which produces 8.4 MW of power. The effluent (exiting flow) from the high-pressure turbine enters the low-pressure turbine at 1 MPa and 250C and leaves at 120 kPa with a quality of 94%. Units f. Ref Phase T P V h Steam P = 15 MPa T = 600C u 5.MP=8.4 MW HI-P Turbine 1 a. Fill in the table of thermodynamic properties you need for this problem. b. Draw p-v and T-v diagrams in the indicated spaces. c. Is it appropriate to use the ideal gas law for the entering steam? [find Z] d. State the assumptions you use to solve this problem e. Find the mass flow rate of steam in kg/s Determine the total power output of the two-stage turbine system in MW. Q=0 MW 2 P = 1 MPa T, = 250C Low-P Turbine Q=0 MW 3 ??? MW P = 120 kPa x = 0.94 kg vap/kg TA 1) Consider the two-stage, adiabatic steam turbine, shown below. Steam at 15 MPa and 600C enters the high-pressure turbine, which produces 8.4 MW of power. The effluent (exiting flow) from the high-pressure turbine enters the low-pressure turbine at 1 MPa and 250C and leaves at 120 kPa with a quality of 94%. Units f. Ref Phase T P V h Steam P = 15 MPa T = 600C u 5.MP=8.4 MW HI-P Turbine 1 a. Fill in the table of thermodynamic properties you need for this problem. b. Draw p-v and T-v diagrams in the indicated spaces. c. Is it appropriate to use the ideal gas law for the entering steam? [find Z] d. State the assumptions you use to solve this problem e. Find the mass flow rate of steam in kg/s Determine the total power output of the two-stage turbine system in MW. Q=0 MW 2 P = 1 MPa T, = 250C Low-P Turbine Q=0 MW 3 ??? MW P = 120 kPa x = 0.94 kg vap/kg TA
Expert Answer:

Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these mechanical engineering questions
-
The T-s diagram of an ideal Rankine power cycle with two reheat cycles and two open feedwater heaters is shown in Fig. 8.39. Steam leaves the boiler at state 7 at 15 MPa and 600°C. Steam at 4 MPa...
-
A steam power plant operates on an ideal reheat- regenerative Rankine cycle with one reheater and two feedwater heaters, one open and one closed. Steam enters the high-pressure turbine at 15 MPa and...
-
Ethanol reacts with sodium and forms two products which are Options: 1) Sodium ethanoate and hydrogen 2) Sodium ethanoate and oxygen 3) Sodium ethoxide and hydrogen 4) Sodium ethoxide and oxygen
-
The records of ZZ Hat Company showed sales revenue of $100,000 (on the income statement) and a change in the balance of accounts receivable. To demonstrate the effect of changes in accounts...
-
Organic Chemistry Structure and Function by Peter Vollhardt and Neil Schore 6th Edition, exercise 19-9. Someone please teach me. Exercise 19-9 Try It Yourself Using chemical equations, show how you...
-
How many ways can a company select 4 candidates to interview from a short list of 12 engineers?
-
Bonds Company purchased a new plant asset on April 1, 2014, at a cost of $355,500. It was estimated to have a service life of 20 years and a salvage value of $30,000. Bondss accounting period is the...
-
On January 1 of Year 1, Alpha Corporation issued $400,000 of 8% (cash payable semiannually on June 30 and December 31), three-year bonds to yield 10%. Required a. Compute the selling price of the...
-
isabelle sterling, the partner you work for, unexpectedly had to travel to Hong Kong. She left you the Tang Aluminum Products file for your client, Global Bank. Global will be lending Tang 1.9...
-
You need to write about Web Field Trip Target, Google, and Whole Foods 1. How well does the strategy fit the company's situation? 2. Is the strategy helping the company achieve a sustainable...
-
A glass rod rubbed against silk gains a charge of -7.4 C. What is the electric force between the rod and the silk when the two are separated by a distance of 11.6 cm? (Assume that the charges are...
-
A box weighing 408 N is hung from two cables. The angle between horizontal and each cable is 32.5. What is the tension in each cable? N
-
Three students are pulling on ropes attached to a ring as shown in the figure below. Determine the magnitude and direction of the net force. Enter the direction as an angle measured in degrees...
-
This figure shows a uniform electric field of E and a surface perpendicular to this field. What is the electric flux through the 5 cm by 5 cm surface? E E = 142 N/C O a. 3550.00 N m^2/C O b. 0.35 N...
-
Q6. Two line charges with uniform charge density pl are sitting on the x-y plane as shown in figure. Determine the electric field intensity on the z-axis. -b Z P(0, 0, z) -a f a b
-
List five reasons why organizations outsource. When should an organization choose not to outsource? Why are some organizations moving their software development work back in house? Why are some...
-
Make an argument that Williams had a right to delay the closing until after August 1.
-
Use the differential form for dP (see Eq. 7.7) assuming P = RT/ v and find the change in the pressure of air if the specific volume and temperature change from 0.5 m3/kg and 80 C to 0.52 m3/kg and 70...
-
Rework Problem 9.43 except that the pressure ratio and maximum temperature are, respectively, a) 4 and 1200°C, b) 5 and 1400°C, c) 6 and 1600°C. Air enters the ideal regenerative Brayton...
-
Find a relationship for Cp - Cp for a gas for which the van der Waals equation of state P = RT / (v - b) - a/v2 is applicable. Then let a = b = 0 and show that Cp - Cv = R.
-
Explain the working of domestic refrigeration system with a neat sketch?
-
Fluid is a substance that: (a) Cannot be subjected to shear force (b) Always expands until it fills any container (c) Has the same shear stress at a point regardless of its motion (d) Cannot remain...
-
Discuss about psychrometry and their properties?

Study smarter with the SolutionInn App


