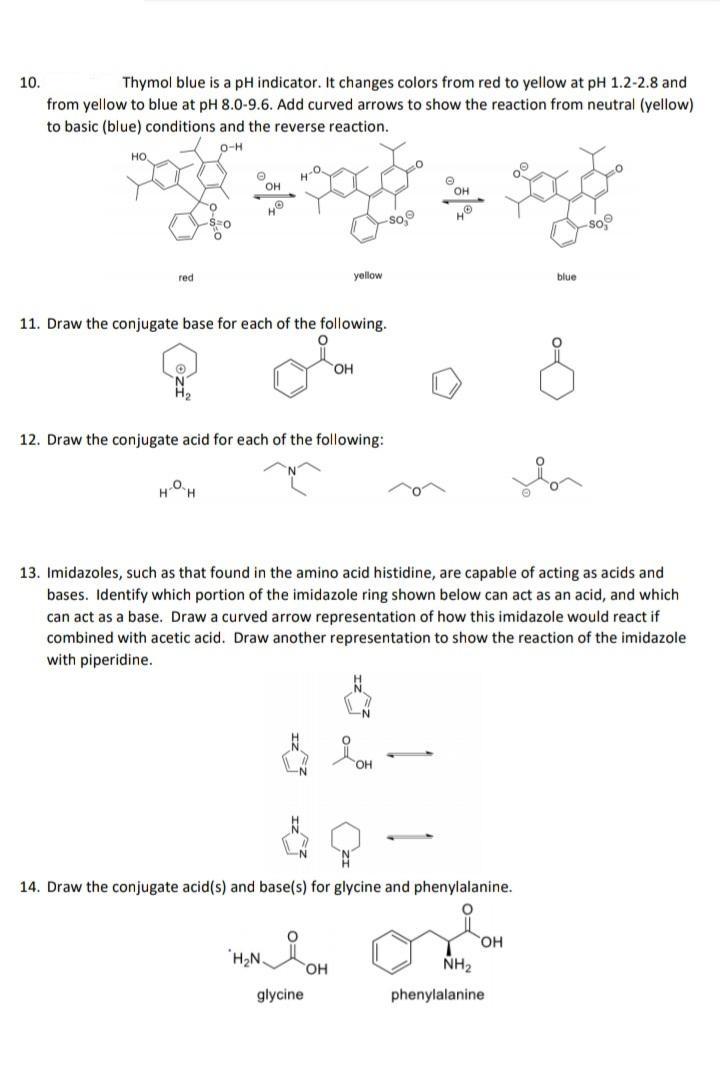
10. Thymol blue is a pH indicator. It changes colors from red to yellow at pH...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
10. Thymol blue is a pH indicator. It changes colors from red to yellow at pH 1.2-2.8 and from yellow to blue at pH 8.0-9.6. Add curved arrows to show the reaction from neutral (yellow) to basic (blue) conditions and the reverse reaction. O-H HO © OH soco HO HO So, -So red yellow blue 11. Draw the conjugate base for each of the following. OH 12. Draw the conjugate acid for each of the following: H-O-H 13. Imidazoles, such as that found in the amino acid histidine, are capable of acting as acids and bases. Identify which portion of the imidazole ring shown below can act as an acid, and which can act as a base. Draw a curved arrow representation of how this imidazole would react if combined with acetic acid. Draw another representation to show the reaction of the imidazole with piperidine. N N 요. OH -N N 14. Draw the conjugate acid(s) and base(s) for glycine and phenylalanine. OH H₂Ni NH₂ glycine phenylalanine O -S=0 OH 10. Thymol blue is a pH indicator. It changes colors from red to yellow at pH 1.2-2.8 and from yellow to blue at pH 8.0-9.6. Add curved arrows to show the reaction from neutral (yellow) to basic (blue) conditions and the reverse reaction. O-H HO © OH soco HO HO So, -So red yellow blue 11. Draw the conjugate base for each of the following. OH 12. Draw the conjugate acid for each of the following: H-O-H 13. Imidazoles, such as that found in the amino acid histidine, are capable of acting as acids and bases. Identify which portion of the imidazole ring shown below can act as an acid, and which can act as a base. Draw a curved arrow representation of how this imidazole would react if combined with acetic acid. Draw another representation to show the reaction of the imidazole with piperidine. N N 요. OH -N N 14. Draw the conjugate acid(s) and base(s) for glycine and phenylalanine. OH H₂Ni NH₂ glycine phenylalanine O -S=0 OH
Expert Answer:

Related Book For 

Organic Chemistry A Short Course
ISBN: 978-1111425562
13th edition
Authors: Harold Hart, Christopher M. Hadad, Leslie E. Craine, David J. Hart
Posted Date:
Students also viewed these chemistry questions
-
Draw the conjugate base for each of the following acids: (a) (b) (c) NH 3 (d) H 3 O + (e) (f) (g) (h) NH 4 + .
-
Draw the conjugate acid for each of the following bases: (a) (b) (c) NaNH 2 (d) H 2 O (e) (f) (g) (i) NaOH
-
Draw perspective representation for each of the following chiral molecules, Use models if necessary, (D = deuterium = 2H, a heavy isotope of hydrogen.) (2Z,4R) - 4 - methyl - 2 - hexane
-
The operations of Balloons Ltd. generated the following data for its current December 31 taxation year: Business losses (150,000) Dividends received and deducted (Division C) 25,000 Bond interest...
-
What amount must be remitted if the following invoices, all with terms 4/10, 2/30, n/60, are paid on May 10? $850 less 20% and 10% dated March 21 $960 less 30 and 16 23% dated April 10 $1040 less 33...
-
What is a database?
-
Describe the shape of the distribution. Use the data set, which represents the points scored by each player on the Montreal Canadiens in the 20152016 NHL season. 7507 17 10 0 19 2 18 9 5 5 26 26 0 12...
-
The Midwest Division of the Paibec Corporation manufactures subassemblies that are used in the corporations final products, Lynn Hardt of Midwests Profit Planning Department has been assigned the...
-
In the last column for NYTD,figure out how NYT'sDNA elements will impact their performance. What DNA elements should NYTD keep, what should it jettison, and what should it change? A element...
-
Apollo Shoes is an audit case designed to introduce you to the entire audit process, from planning the engagement to drafting the final report. You are asked to assume the role of a veteran of...
-
Influencer Marketing is big business. Have you ever been influenced to buy a product or service by an online influencer? Were you happy with the product/experience? Do you feel more or less trust in...
-
For the following products and countries, identify the type of warehouse that should be used as well as the method of transportation that should deliver the product to end users. Using the Internet,...
-
Consider switching from one large chiller (1,000 ton) to four chillers (each rated at 250 ton). Using the normalized savings shown in Figure 8.10, estimate the annual energy savings for using a multi...
-
How many axes of reflection symmetry does the triangle in Figure P1.6 have? Restrict your answer to axes that lie in the plane of the triangle. Figure P1.6
-
a. Write the \(\mathrm{AR}(1)\) error model \(e_{t}=ho e_{t-1}+v_{t}\) in lag operator notation. b. Show that and hence that (1pL)=1+ pL + pL + pL +... -
-
Three identical coins are positioned on a grid as shown in Figure P1.8. Where would you place a fourth coin to form a coin arrangement that has both reflection symmetry and a 90 rotational symmetry?...
-
Indirect Costs on a project are Rs. 50/day. A can be crashed by 3 days at a cost of Rs. 40/day. B can be crashed by 4 days at a cost of Rs. 60/day. What is the optimal strategy? 1.Crash A by 3 days...
-
For the following arrangements, discuss whether they are 'in substance' lease transactions, and thus fall under the ambit of IAS 17.
-
What features will be similar in the IR spectra of the following compounds, and how will their IR spectra differ? H2C CHCHCH and CH CH CHCH,CHCH sDh
-
Mutations (caused by radiation, cancer-producing agents, or other means) may replace one base with another or may add or delete a base. What would happen to the protein produced if the sequence UUU...
-
Show the products expected from complete hydrolysis of Gly-Ala-Ser.
-
What are the basic forms of international business activity?
-
Discuss the meaning of international business.
-
What is international business? How does it differ from domestic business?

Study smarter with the SolutionInn App


