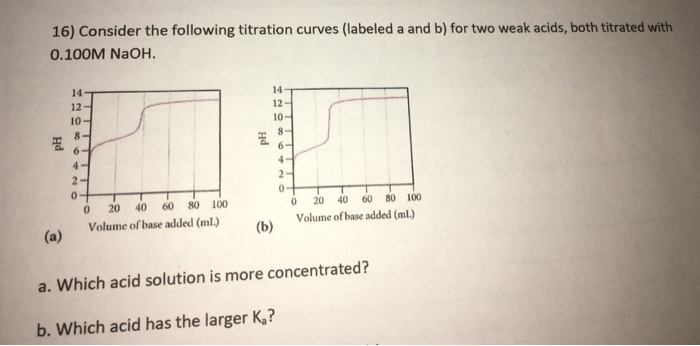
16) Consider the following titration curves (labeled a and b) for two weak acids, both titrated...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
16) Consider the following titration curves (labeled a and b) for two weak acids, both titrated with 0.100M NaOH. Hd 14 12 10- 8. 6- 4- 2- 0 20 40 60 80 100 Volume of base added (ml.) 0 Hd (b) 14- 12- 10- 8 6- 4 2- 0- 20 40 60 80 100 Volume of base added (mL.) 0 a. Which acid solution is more concentrated? b. Which acid has the larger K₂? 16) Consider the following titration curves (labeled a and b) for two weak acids, both titrated with 0.100M NaOH. Hd 14 12 10- 8. 6- 4- 2- 0 20 40 60 80 100 Volume of base added (ml.) 0 Hd (b) 14- 12- 10- 8 6- 4 2- 0- 20 40 60 80 100 Volume of base added (mL.) 0 a. Which acid solution is more concentrated? b. Which acid has the larger K₂?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following supply and demand schedule for steel: Price per tonne ($) 20 40 60 80 100 120 140 160 180 Q demanded (million tonnes) 200 180 160 140 120 100 80 60 40 Q supplied (million...
-
We have vector x = [20 40 60 80 100 120 140 160 180 200] and we want to multiply all the elements of the vector x e.g. (20 x 40 x 60 . x 200) and store the result in temp. You can use it for the loop.
-
Find the equivalent resistance between points A and B for the group of resistors shown in FIGURE 21-40. wwww 35 0 82 0 45 0
-
The comparative balance sheet of Beets Industries, Inc. at December 31, 2013 and 2012, is as follows: An examination of the income statement and the accounting records revealed the following...
-
What is the forward rate for year 3 (the forward rate quoted today for an investment that begins in two years and matures in three years)? What can you conclude about forward rates when the yield...
-
Malitz Inc. recently hired you as a consultant to estimate the companys WACC. You have obtained the following information. (1) Malitzs no callable bonds mature in 25 years, have an 8.00% annual...
-
A company wishes to hedge its exposure to a new fuel whose price changes have a 0.6 correlation with gasoline futures price changes. The company will lose $1 million for each 1 cent increase in the...
-
On September 1, Roshek Office Supply had an inventory of 30 calculators at a cost of $22 each. The company uses a perpetual inventory system. During September, the following transactions occurred....
-
2. Please using k-means clustering method to cluster these data points in to 2 clusters. Please provide step by step operations. (10 points) Sample: Feature 1 Feature Step1: 1 1234567 2.5 1.5 2 2.0...
-
In the light of this analysis, what strategic options would you advise a Western oil producer such as Shell or BP to consider?
-
3. a. For set-Associative mapping if Main Memory: 64GB, Block/Line Size: 16B, Cache:512 KB with 4- way set associative: [4] i. How many address bits are required to address each byte location in RAM?...
-
Define P(n) to be the assertion that: (a) Verify that P(3) is true. (b) Express P(k) (c) IM- n(n + 1)(2n+1) 6 Express P(k + 1). (d) In an inductive proof that for every positive integer n...
-
How to calculate a 3-month spot rate as an annual rate with monthly compounding. Zero coupon bond Maturity Month Bond Price 3 99.194 6 98.225 9 97.454 12 95.807 15 94.238 18 92.873 21 90.236 24 88.096
-
2. From the items listed below, what amount of allowable deductions can be subtracted from the gross estate? Funeral expenses $4,000 Executor's fees 5,800 Mortgage on tenants in common property (20%...
-
a. If you are comparing three or more groups what nonparametric test statistics is more to use and why? b. what procedures we are going to do if we want to know which pair groups are different?
-
A capacitor is built of two non-parallel, rectangular metallic plates. In a cross-sectional view the capacitor is composed of two right-angled triangles with differently sized sides (a,>a,), but they...
-
The system shown in the figure is how much Q force is obtained at the output for an input of P = 1000 N? Find the bearing and joint forces.
-
A sample statistic will not change from sample to sample. Determine whether the statement is true or false. If it is false, rewrite it as a true statement.
-
Draw short segments of the polymers obtained from the following monomers: a.CH2=CHF b. CH2=CHCO2H c. d. e. HOCH2sCOH NCOHOCH CH OH OCN
-
Give the major product obtained from reaction of each of the following with excess HCl: a. CH3CH2C==CH b. CH3CH2C===CCH2CH3 c. CH3CH2C==CCH2CH2CH3
-
The mechanism for hydroxide-ion-promoted ester hydrolysis is shown in Section 17.12. What catalytic role does hydroxide ion play in this mechanism?
-
Consider the electrical circuit shown below where \(v_{i}(t)=\beta\), for \(t \geq 0\). Find the dynamic system response \(v_{o}(t)\). VC C ww R V;(t) R ww R3 A First-Order Electrical System Vo (t) +
-
Find the dynamic system response for the spring mass damping system shown in Figure 3.2, where \(f(t)=\beta, x(0)=0\) (i.e. there is no initial energy stored). b f(t) k Figure 3.2 A first-order...
-
Consider a first-order system with a forcing function \(f(t)\) such that input-output differential equation is given by \[\begin{equation*}\dot{y}+3 y=f(t) \text {, } \tag{3.7}\end{equation*}\] where...

Study smarter with the SolutionInn App



