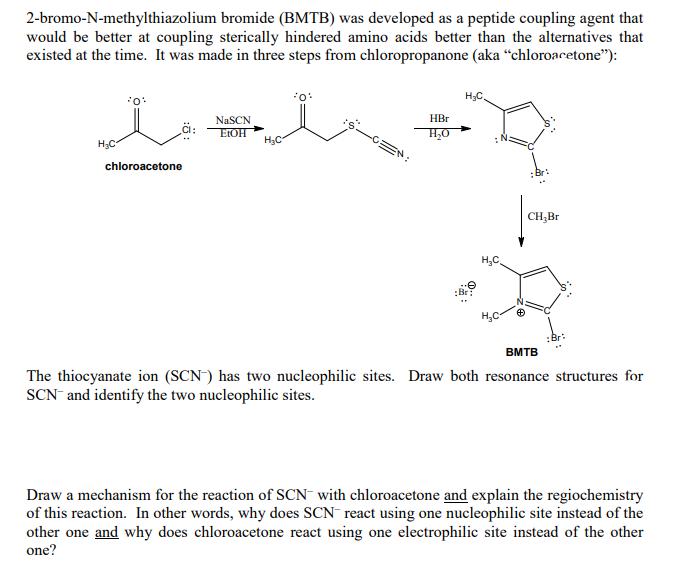
2-bromo-N-methylthiazolium bromide (BMTB) was developed as a peptide coupling agent that would be better at coupling...
Fantastic news! We've Found the answer you've been seeking!
Question:
![]()
Transcribed Image Text:
2-bromo-N-methylthiazolium bromide (BMTB) was developed as a peptide coupling agent that would be better at coupling sterically hindered amino acids better than the alternatives that existed at the time. It was made in three steps from chloropropanone (aka "chloroacetone"): H₂C *0* CI: chloroacetone NaSCN EtOH H₂C *0* HBr H₂O H₂C :Br: H₂C CH₂Br ;Br: BMTB The thiocyanate ion (SCN) has two nucleophilic sites. Draw both resonance structures for SCN and identify the two nucleophilic sites. Draw a mechanism for the reaction of SCN with chloroacetone and explain the regiochemistry of this reaction. In other words, why does SCN react using one nucleophilic site instead of the other one and why does chloroacetone react using one electrophilic site instead of the other one? (c) Propose a reasonable mechanism for the second step in the synthesis of BMTB. (shown below) 2-bromo-N-methylthiazolium bromide (BMTB) was developed as a peptide coupling agent that would be better at coupling sterically hindered amino acids better than the alternatives that existed at the time. It was made in three steps from chloropropanone (aka "chloroacetone"): H₂C *0* CI: chloroacetone NaSCN EtOH H₂C *0* HBr H₂O H₂C :Br: H₂C CH₂Br ;Br: BMTB The thiocyanate ion (SCN) has two nucleophilic sites. Draw both resonance structures for SCN and identify the two nucleophilic sites. Draw a mechanism for the reaction of SCN with chloroacetone and explain the regiochemistry of this reaction. In other words, why does SCN react using one nucleophilic site instead of the other one and why does chloroacetone react using one electrophilic site instead of the other one? (c) Propose a reasonable mechanism for the second step in the synthesis of BMTB. (shown below)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw both resonance structures of the enolate formed when each of the following ketones is treated with a strong base: (a) (b) (c) (d) (e)
-
Are amino acids other than the usual 20 amino acids found in proteins? If so, how are such amino acids incorporated into proteins? Give an example of such an amino acid and a protein in which it...
-
Propose a mechanism for the reaction of (a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane. (b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane?
-
We consider the following CFG SE + SIE E 01|2|3|4|5|67|8|9|(S) Apply the leftmost derivation and rightmost derivation with a top-down parser for this sentence: (3+7+ (1+4)) + 2
-
Fuqua Company has accumulated the following budget data for the year 2010. 1. Sales: 30,000 units, unit selling price $80. 2. Cost of one unit of finished goods: Direct materials 2 pounds at $5 per...
-
What is the function of the mitotic spindle?
-
For each of the definitions, write the letter of the appropriate term in the space provided. 1_The logical sequence in which the computer expects each text box, option button, or command to be...
-
On January 20, Metropolitan, Inc., sold 8 million shares of stock in an SEO. The market price of Metropolitan at the time was $42.50 per share. Of the 8 million shares sold, 5 million shares were...
-
Explain TEN (10) ways in which an Accounting Information System can add value to an organization.
-
Successful completion of a construction project depends on the supply of materials and labor as well as the weather condition. Consider a given project that can be success fully completed if either...
-
What protection is given to the owner of a patent?
-
if they want to pay $12,500 per year for fours years to pay for Jon's college, how much do they need to fund that. he is 16 years old and will go go college at age 18 for four years. If they started...
-
What are the latest advancements in process intensification for reducing reactor size while maintaining high production rates, and how do they impact sustainability in chemical manufacturing ?
-
The Corner Art Gallery purchased a painting for $68,000 two years ago. Today, the painting was resold for $179,900. What annual rate of return did the gallery earn on this painting?
-
2. Consider an infinite sheet of charge with charge density in the z - 0 direction with a speed v: I K y plane. The charge uniformly moves in the y Y You can think of this as a steady surface current...
-
9. A consumer electronics company was formed to develop cell phones that run on or are recharged by fuel cells. The company purchased a warehouse and converted it into a manufacturing plant for...
-
Which of the following statements is true regarding discount notes?
-
Why is the national security argument for tariffs questionable?
-
Beleaguered State Bank (BSB) holds $250 million in deposits and maintains a reserve ratio of 10 percent. a. Show a T-account for BSB. b. Now suppose that BSBs largest depositor withdraws $10 million...
-
Using a diagram of the labor market, show the effect of an increase in the minimum wage on the wage paid to workers, the number of workers supplied, the number of workers demanded, and the amount of...
-
What kind of agreement is illegal for businesses to make? Why are the antitrust laws controversial?
-
Like many American cities during the 1960s and 1970s, Washington, DC, experienced declining population, economic challenges, and growing social problems, as many middle-class families and businesses...
-
Think about your most rewarding (or unrewarding) experience as a volunteer or nonprofit staff member. Which of Herzberg's satisfiers and dissatisfiers were present in the environment?
-
Are the values and motivations of millennials really different from those of previous generations or do people become more alike as they proceed through various life stages?

Study smarter with the SolutionInn App



