3. (30 points) A sample of pure helium gas at 20.0C and a pressure of 6.0...
Fantastic news! We've Found the answer you've been seeking!
Question:

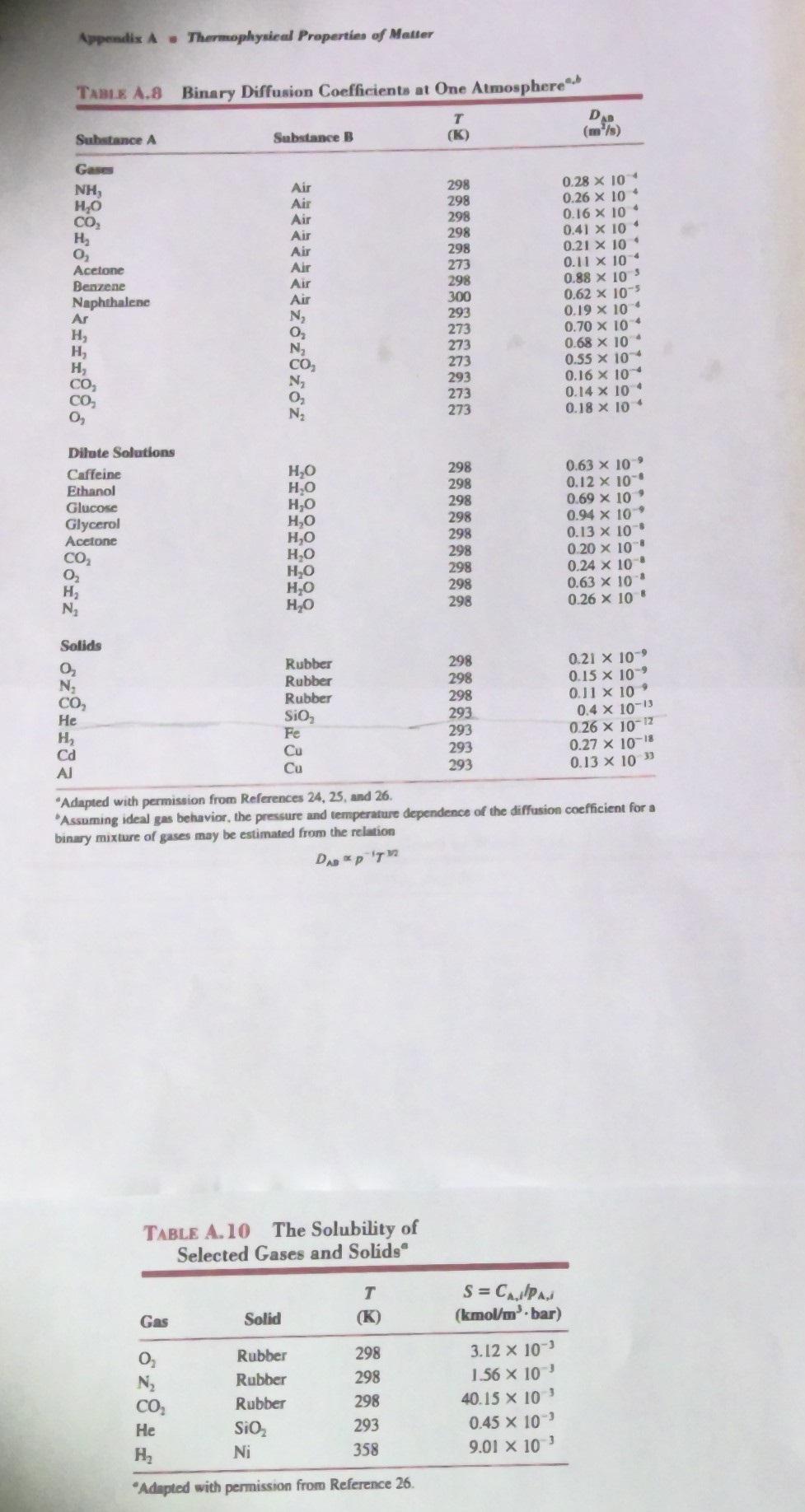
Transcribed Image Text:
3. (30 points) A sample of pure helium gas at 20.0C and a pressure of 6.0 bars is stored in a glass (SiO2) cylindrical vial with an inner diameter of 25 mm and a wall thickness of 1.00 mm. There is negligible helium at the exterior vial surface. We can assume the helium gas is an ideal gas. Assume diffusion is one-dimensional through the thickness of the vial wall. Analyze the system assuming steady state. There is no generation or consumption of He in the glass. Assume the total molar concentration C is constant and He is dilute in the vial wall. He gas 12 (a) Use Tables A.8 and A. 10 to find the diffusivity and solubility of helium gas in the glass. (b) Using the solubility, determine the helium molar concentration (CA) at the interior surface of the glass (r = r;). What is the helium concentration at the exterior glass surface (r = r)? (e) Starting from the governing Mass Diffusion Equation and using Fick's Law, find the simplified MDE in terms of the helium molar concentration (CA) in the vial glass wall (d) Solve the simplified equation in (e) for the general solution of the concentration profile C(r) in the glass wall of the vial (e) Solve for the symbolic expression of the helium concentration in the vial wall, C^(r) (f) What is the mass transfer rate of helium per unit height (L) of the vial in g/(m/s)? Appendix A Thermophysical Properties of Matter TABLE A.8 Binary Diffusion Coefficients at One Atmosphere T Substance A Substance B (K) DAB (m/s) Gases NH, Air 298 0.28 x 10 HO Air 298 CO Air 298 0.26 x 10 * 0.16 x 10 H Air 298 0.41 x 104 0 Air 298 0.21 x 10' Acetone Air 273 0.11 10 Benzene Air 298 0.88 x 105 Naphthalene Air 300 0.62 x 105 Ar N 293 0.19 10 4 H 0 273 0.70 x 10 H N 273 0.68 x 10 H CO 273 0.55 10 CO N 293 0.16 10 CO 273 0.14 x 104 0 N 273 0.18 10 4 Dilute Solutions Caffeine HO 298 0.63 x 109 Ethanol HO 298 0.12 10 Glucose Glycerol Acetone HO 298 0.69 x 10' HO 298 0.94 x 10' HO 298 0.13 x 10 8612 HO 298 0.20 x 10 HO 298 0.24 x 10 HO 298 0.63 x 10' HO 298 0.26 x 10" Solids 0 Rubber 298 0.21 10 N Rubber 298 0.15 x 109 CO Rubber 298 0.11 x 10' He SiO 293 0.4 10-13 H Cd Al 233 Fe 293 0.26 10-12 Cu 293 0.27 x 10-18 293 0.13 x 10" "Adapted with permission from References 24, 25, and 26. Assuming ideal gas behavior, the pressure and temperature dependence of the diffusion coefficient for a binary mixture of gases may be estimated from the relation DAB PT TABLE A.10 The Solubility of Selected Gases and Solids T Gas Solid (K) S = CAPA (kmol/m bar) 0 Rubber 298 3.12 x 10 N Rubber 298 1.56 x 10 CO Rubber 298 40.15 x 10' He SiO 293 0.45 x 10' H Ni 358 9.01 10 "Adapted with permission from Reference 26. 3. (30 points) A sample of pure helium gas at 20.0C and a pressure of 6.0 bars is stored in a glass (SiO2) cylindrical vial with an inner diameter of 25 mm and a wall thickness of 1.00 mm. There is negligible helium at the exterior vial surface. We can assume the helium gas is an ideal gas. Assume diffusion is one-dimensional through the thickness of the vial wall. Analyze the system assuming steady state. There is no generation or consumption of He in the glass. Assume the total molar concentration C is constant and He is dilute in the vial wall. He gas 12 (a) Use Tables A.8 and A. 10 to find the diffusivity and solubility of helium gas in the glass. (b) Using the solubility, determine the helium molar concentration (CA) at the interior surface of the glass (r = r;). What is the helium concentration at the exterior glass surface (r = r)? (e) Starting from the governing Mass Diffusion Equation and using Fick's Law, find the simplified MDE in terms of the helium molar concentration (CA) in the vial glass wall (d) Solve the simplified equation in (e) for the general solution of the concentration profile C(r) in the glass wall of the vial (e) Solve for the symbolic expression of the helium concentration in the vial wall, C^(r) (f) What is the mass transfer rate of helium per unit height (L) of the vial in g/(m/s)? Appendix A Thermophysical Properties of Matter TABLE A.8 Binary Diffusion Coefficients at One Atmosphere T Substance A Substance B (K) DAB (m/s) Gases NH, Air 298 0.28 x 10 HO Air 298 CO Air 298 0.26 x 10 * 0.16 x 10 H Air 298 0.41 x 104 0 Air 298 0.21 x 10' Acetone Air 273 0.11 10 Benzene Air 298 0.88 x 105 Naphthalene Air 300 0.62 x 105 Ar N 293 0.19 10 4 H 0 273 0.70 x 10 H N 273 0.68 x 10 H CO 273 0.55 10 CO N 293 0.16 10 CO 273 0.14 x 104 0 N 273 0.18 10 4 Dilute Solutions Caffeine HO 298 0.63 x 109 Ethanol HO 298 0.12 10 Glucose Glycerol Acetone HO 298 0.69 x 10' HO 298 0.94 x 10' HO 298 0.13 x 10 8612 HO 298 0.20 x 10 HO 298 0.24 x 10 HO 298 0.63 x 10' HO 298 0.26 x 10" Solids 0 Rubber 298 0.21 10 N Rubber 298 0.15 x 109 CO Rubber 298 0.11 x 10' He SiO 293 0.4 10-13 H Cd Al 233 Fe 293 0.26 10-12 Cu 293 0.27 x 10-18 293 0.13 x 10" "Adapted with permission from References 24, 25, and 26. Assuming ideal gas behavior, the pressure and temperature dependence of the diffusion coefficient for a binary mixture of gases may be estimated from the relation DAB PT TABLE A.10 The Solubility of Selected Gases and Solids T Gas Solid (K) S = CAPA (kmol/m bar) 0 Rubber 298 3.12 x 10 N Rubber 298 1.56 x 10 CO Rubber 298 40.15 x 10' He SiO 293 0.45 x 10' H Ni 358 9.01 10 "Adapted with permission from Reference 26.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Which of the following procedures most likely represents an internal control designed to reduce the risk of errors in the billing process? 1. Requiring customers that purchase on account to be...
-
Deborah purchases a new $30,000 car in 2013 to use exclusively in her business. If Deborah does not elect to expense or take bonus depreciation in 2013 and holds the car until it is fully...
-
Use a triple integral to find the volume of the given solid. The solid enclosed by the cylinder x 2 + z 2 = 4 and the planes y = -1 and y + z = 4
-
Political expectations, inflation, and unemployment Consider a country with two political parties, Democrats and Republicans. Democrats care more about unemployment than Republicans, and Republicans...
-
The intangible assets section of Glover Company at December 31, 2013, is presented below. Patents ($60,000 cost less $6,000 amortization) ....... $54,000 Franchises ($48,000 cost less $19,200...
-
A company is planning to purchase and store two items, gadgets and widgets. Each gadget costs $2.00 and occupies 2 square meters of floor space; each widget costs $3.00 and occu pies 1 square meter...
-
What is the price of a bond that has a twelve-year maturity, a coupon rate of 3.45 percent, a par value of $1,000, and pays interest semiannually? The current market yield is 4.56 percent.
-
13. Bob takes a bathroom scale into an elevator. (A bathroom scale measures the normal force it must exert to support your weight- that's why it shifts if you shift your weight while standing on it.)...
-
A small amount of radioactive solution containing the isotope 24Na is injected to a patient in a hospital. The activity of the solution at the injection moment is Ao = 2000 Bq. After 5 hours the...
-
Consider two vectors, N, shown in the picture and vector Mof magnitude 4.1 cm that makes an angle of 240 degrees (measured counterclockwise) with +x axis. What is the magnitude of the sum of these...
-
Given a capacitor of C = 11 F, calculate the current going through the capacitor during the interval t2
-
1. For each case determine the net forces acting upon the object and determine its direction Situation A Ffrict=5 N = 3 N Fnorm Fapp = 5 N Fgray = 3 N Situation B Ffrict = 5 N = 3 N norm Fgray = 3 N
-
Zina Chaburi has a full time job as a nurse in her local hospital. In her spare time she has agoat farming operation. The goat farm began in 2013, and resulted in a loss of $10,000.She deducted the...
-
Sportique Boutique reported the following financial data for 2012 and 2011. Instructions(a) Calculate the current ratio for Sportique Boutique for 2012 and 2011.(b) Suppose that at the end of 2012,...
-
A certain metal M forms a bromide containing 53.79 percent Br by mass. What is the chemical formula of the compound?
-
Deduce plausible monomers for polymers with the following repeating units: (a) (b) + CH-_-CF, CONH
-
The following diagram represents the products (CO2 and H2 O) formed after the combustion of a hydrocarbon (a compound containing only C and H atoms). Write an equation for the reaction. CO2 H20
-
Fill in the blanks to make the following statements correct. a. Economists have designed____________ to better explain and predict the behaviour we observe in the world around us. b. A variable, such...
-
The following supply and demand schedules describe a hypothetical Canadian market for potash. a. What is the equilibrium price of potash? b. How much potash would actually be purchased if the price...
-
According to Statistics Canada, Canada's exports and imports of energy (combined totals of fossil fuels, hydro, and nuclear, all measured in petajoules) over a five-year period were as follows: a....

Study smarter with the SolutionInn App


