5. Calculate the overvoltage (in V) that is needed to sustain a current density of 20...
Fantastic news! We've Found the answer you've been seeking!
Question:
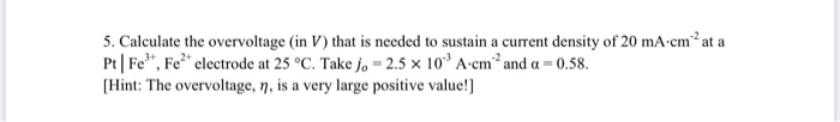
Transcribed Image Text:
5. Calculate the overvoltage (in V) that is needed to sustain a current density of 20 mA-cm² at a Pt| Fe", Fe" clectrode at 25 °C. Take jo = 2.5 x 10 A-cm and a = 0.58. (Hint: The overvoltage, n, is a very large positive value!] 5. Calculate the overvoltage (in V) that is needed to sustain a current density of 20 mA-cm² at a Pt| Fe", Fe" clectrode at 25 °C. Take jo = 2.5 x 10 A-cm and a = 0.58. (Hint: The overvoltage, n, is a very large positive value!]
Expert Answer:
Answer rating: 100% (QA)
Given current density of 20 mAcm 3 Temperature 25 degrees celsius or 25273298 K To find overvolt... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What over potential is needed to sustain a current of 15 mA at a Pt] Ce4+'Ce3+ electrode in which both ions are at a mean activity a = 0.01D?
-
A current density of 6.00 x 10-13 A/m2 exists in the atmosphere at a location where the electric field is 100 V/m. Calculate the electrical conductivity of the Earths atmosphere in this region.
-
A current density of 0.05 A/cm2 is applied to a 150 cm2 cathode. What period of time is required to plate out a 1-mm-thick coating of silver onto the cathode?
-
Gagnon's Autobody Ltd. repairs and paints automobiles after accidents. Explain how the basic statement of financial position accounts of assets, liabilities, and shareholders' equity would be...
-
A bismuth-209 nucleus reacts with an alpha particle to produce an astatine nucleus and two neutrons. Write the complete nuclear equation for this reaction.
-
The adjusted trial balance for Tsai Bowling Alley at December 31, 2025, contains the following accounts (amounts in thousands). Instructions a. Prepare a classified statement of financial position;...
-
The following readings were taken during a test on a single-cylinder, four-stroke gas engine : Calculate (a) IP, (b) BP, (c) BSFC, (d) brake thermal efficiency, and (e) relative efficiency. Speed of...
-
A project has just completed the 87th item in its plan. It was scheduled to have spent $168,000 at this point in the plan, but has actually spent only $156,000. The foreman estimates that the value...
-
How can conflict theory proposed a solution for crime and education in a community? Provide references!
-
What insights from the discussion of the Morgan Stanley 2003 report on eBay apply to the 2013 report on Aetna by Jefferies? What insights from the discussion of the Morgan Stanley 2003 report on eBay...
-
Find information regarding Target's short and long-term financing, click on 10-K part II and under item #8, select 'Financial Statements and Supplementary Data'. Scroll down to find the Consolidated...
-
Study the organizational chart in figure 3.5. What kinds of day-to-day activities would you expect to take place for the employees in each box? How would you expect them to share information across...
-
What role do focus and reflection play in workplace learning and problem solving?
-
For each number \(x\) in the domain, the corresponding range value \(y\) is found by taking the square root of the domain value subtracted from 5.
-
What types of ethical principles are embedded in the student code of conduct at your university? Should these be followed to the same degree in all student situations? Why or why not?
-
Think about recent graduates of your technical communication program. Where are they employed? What courses and minors did they take with their degrees? Were these courses and minors beneficial to...
-
Large established companies have often difficulties in adapting to technological change. Supercomputer has for a long time been an exception. With a long history of successful R&D and high-quality...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
The data below apply to the formation of urea from ammonium cyanate, NH4CNO --7 NH2CONH2. Initially 22.9 g of ammonium cyanate was dissolved in enough water to prepare 1.00 dm3 of solution. Determine...
-
Suppose that you are a colour chemist and had been asked to intensify the colour of a dye without changing the type of compound, and that the dye in question was a polyene. Would you choose to...
-
Use the Gibbs-Duhem equation to show that the partial molar volume (or any partial molar property) of a component B can be obtained if the partial molar volume (or other property) of A is known for...
-
Vertical analysis would rarely be performed on which of the following statements or schedules? a. Income statement b. Adjusting entry worksheet c. Balance sheet d. All of the above are common targets...
-
A statement that lists the assets, liabilities, and stockholders equity of a company in percentages only with no dollar amounts is a a. common-size income statement. b. benchmarking analysis. c....
-
In vertical analysis, the base used for comparison on the income statement is a. total expenses. b. total assets. c. net sales. d. gross profit.

Study smarter with the SolutionInn App


