Question: 5.5 As shown in Fig. P5.5, a reversible power cycle R and an irreversible power cycle I operate between the same hot and cold
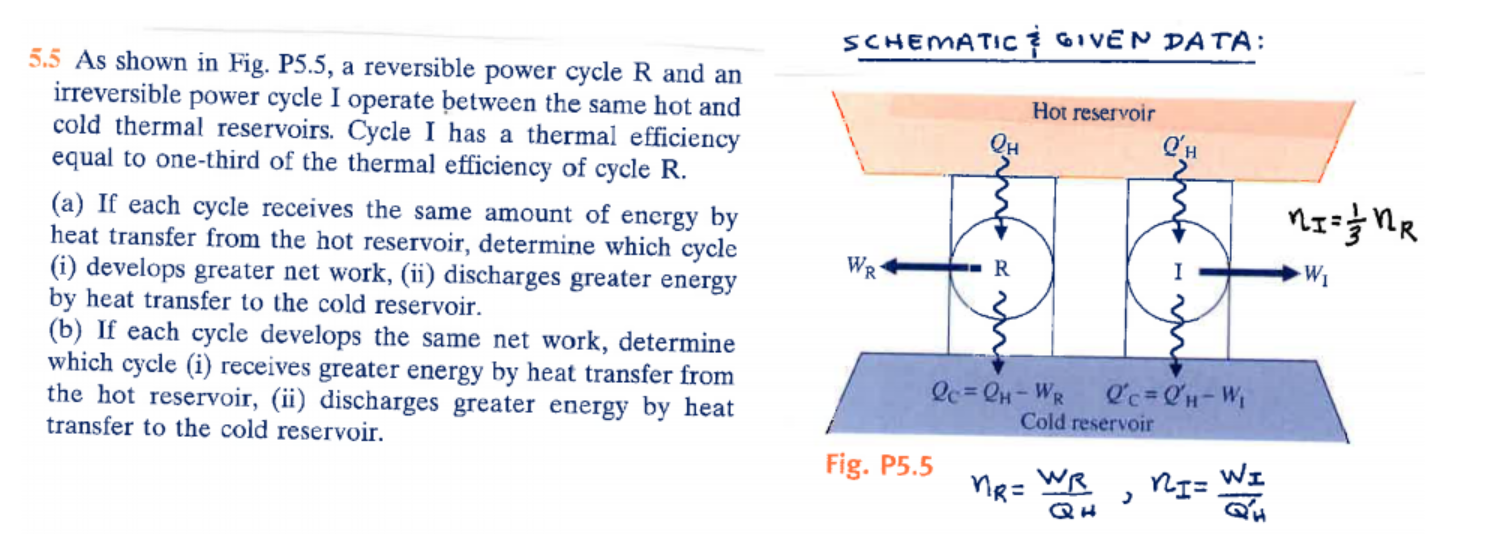
5.5 As shown in Fig. P5.5, a reversible power cycle R and an irreversible power cycle I operate between the same hot and cold thermal reservoirs. Cycle I has a thermal efficiency equal to one-third of the thermal efficiency of cycle R. (a) If each cycle receives the same amount of energy by heat transfer from the hot reservoir, determine which cycle (i) develops greater net work, (ii) discharges greater energy by heat transfer to the cold reservoir. (b) If each cycle develops the same net work, determine which cycle (i) receives greater energy by heat transfer from the hot reservoir, (ii) discharges greater energy by heat transfer to the cold reservoir. SCHEMATIC & GIVEN DATA: WR QH Fig. P5.5 R Hot reservoir Qc=QH-WR l'c=QH-W Cold reservoir nR= WR QH l'H > ni= WI MI = = = nR W
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


