6. Which of the following SN2 reactions is the fastest? A.) CH3CH2CH2CH2BR + OH- B.) CH3CH2CH2CH2B...
Fantastic news! We've Found the answer you've been seeking!
Question:
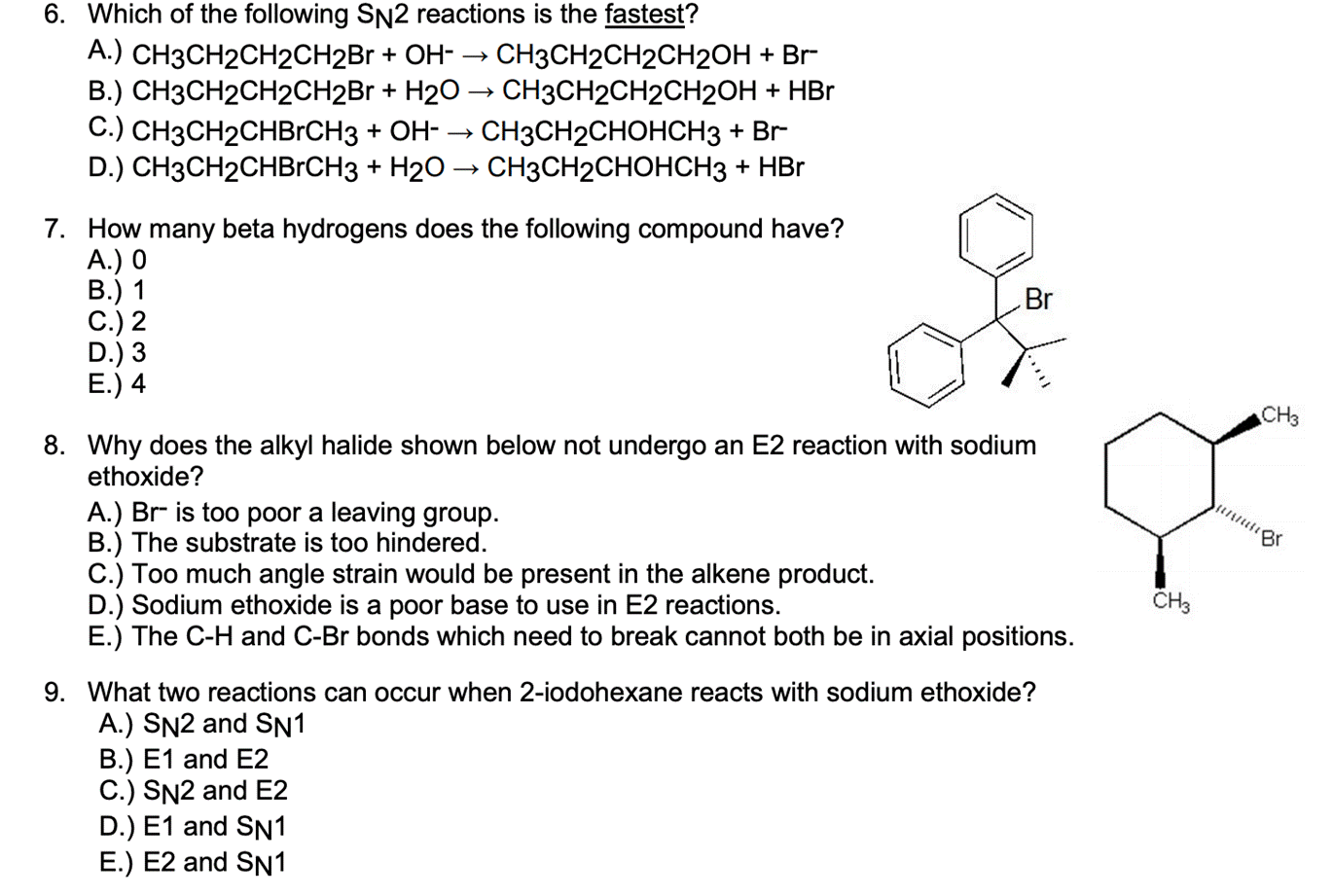
Transcribed Image Text:
6. Which of the following SN2 reactions is the fastest? A.) CH3CH2CH2CH2BR + OH- B.) CH3CH2CH2CH2B + H20 → CH3CH2CH2CH2OH + HBr C.) CH3CH2CHBRCH3 + OH- → D.) CH3CH2CHBRCH3 + H20 → CH3CH2CHOHCH3 + HBr CH3CH2CH2CH2OH + Br CH3CH2CHOHCH3 + Br 7. How many beta hydrogens does the following compound have? A.) 0 В.) 1 C.) 2 D.) 3 E.) 4 Br CH3 8. Why does the alkyl halide shown below not undergo an E2 reaction with sodium ethoxide? A.) Br is too poor a leaving group. B.) The substrate is too hindered. C.) Too much angle strain would be present in the alkene product. D.) Sodium ethoxide is a poor base to use in E2 reactions. E.) The C-H and C-Br bonds which need to break cannot both be in axial positions. 9. What two reactions can occur when 2-iodohexane reacts with sodium ethoxide? A.) SN2 and SN1 B.) E1 and E2 C.) SN2 and E2 D.) E1 and SN1 E.) E2 and SN1 6. Which of the following SN2 reactions is the fastest? A.) CH3CH2CH2CH2BR + OH- B.) CH3CH2CH2CH2B + H20 → CH3CH2CH2CH2OH + HBr C.) CH3CH2CHBRCH3 + OH- → D.) CH3CH2CHBRCH3 + H20 → CH3CH2CHOHCH3 + HBr CH3CH2CH2CH2OH + Br CH3CH2CHOHCH3 + Br 7. How many beta hydrogens does the following compound have? A.) 0 В.) 1 C.) 2 D.) 3 E.) 4 Br CH3 8. Why does the alkyl halide shown below not undergo an E2 reaction with sodium ethoxide? A.) Br is too poor a leaving group. B.) The substrate is too hindered. C.) Too much angle strain would be present in the alkene product. D.) Sodium ethoxide is a poor base to use in E2 reactions. E.) The C-H and C-Br bonds which need to break cannot both be in axial positions. 9. What two reactions can occur when 2-iodohexane reacts with sodium ethoxide? A.) SN2 and SN1 B.) E1 and E2 C.) SN2 and E2 D.) E1 and SN1 E.) E2 and SN1
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
How will the rate of each of the following SN2 reactions change if the polarity of a protic polar solvent is increased? a. b. c. CH,CH2CH2CH2BrHOTCH CH2CH2CH2OHBr CH3 CH,CH31 + NH3 CH3CH,NH, -
-
How many signals would be present in the 1H NMR spectrum of each of the cyclopropane derivatives shown below? Consider carefully the geometric environments around each hydrogen. (a) (b) (c) (d) (e)...
-
What types of marketing opportunities would be present in BRIC countries that would not be available in least developed economies? Would the number of bottom-of-the-pyramid consumers in either...
-
An individual has capital losses brought forward from previous years amounting to 4,800. Compute the individual's taxable gains for 2020-21 if total gains and losses for the year are as follows:...
-
a. "The values of some stocks are dependent on the bond market. When investors are not interested in junk bonds, the values of stocks ripe for leveraged buyouts decline." b. "The recent trend in...
-
Distinguish between a range check and a reasonableness check. Why is a reasonableness check not a field-level check?
-
What is meant by a confluence of results?
-
Acme, Inc., incurs the following costs during May: Sales expense . . . . . . . . . . . . . . . . $ 11,500 Direct labor . . . . . . . . . . . . . . . . . . 26,000 Factory supplies . . . . . . . . . ....
-
An investor has a broadly diversified portfolio of blue chip stocks. The use of index options to hedge the portfolio reduces: A systematic risk B non-systematic risk C interest rate risk D timing risk
-
Suppose you are a managerial accountant or cost analyst for Donut Desire. You have been asked to make a presentation to management about the status of the company's utility costs. Refer to Exhibits...
-
In light of the global COVID-19 pandemic and persistent criticism of the dog meat trade from Chinese citizens, the government of China has reclassified dogs from "livestock" to "companion animals."...
-
From the perspective of organizational structure, design, and control, what have gone wrong at PNB? What factors have contributed to its current state of disarray? What should be done next to help...
-
Your goal is to advise the President on domestic economic policy. Role You are the chair of the Council of Economic Advisors (CEA) Audience Your audience is the President of the United States....
-
omework quiz 2.1 #1 stem plot The miles per gallon rating for 30 cars are shown below (lowest to highest). 19, 19, 19, 20, 21, 21, 25, 25, 25, 26, 26, 28, 29, 31, 31, 32, 32, 33, 34, 35, 36, 37, 37,...
-
Your answers are saved automatically. Question Completion Status: QUESTION 1 13 points Save Answer Library A computer memory manufacturer specifies that its memory chip stores data incorrectly an...
-
Analyze the possible reasons for and responses to Chung's request for a private office. What factors might impact Leary's decision? Identify at least two challenges and dilemmas in managing...
-
9. Bodega Boston's (https://bdgastore.com/) secret shoe retail store has an opportunity to open a store in Barcelona, Spain that will have the same secret store location and sell similar limited...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Draw the enol tautomers for each of the following compounds. For those compounds that have more than one enol tautomer, indicate which is more stable. a. b. c. d. e. f. CH3CH2CCH,CH3 CCH3 CH CH...
-
Show how the following compounds could be prepared, starting with 3 cyanocyclohexanone a. b. CH2CH2CCH
-
Show the aldol addition product that would be formed from each of the following compounds: a. b. c. d. CHCHCH-CHCH - - CH CHCH2CH CH CH3 ,
-
Here are the raw data for the ANES unique group of widows with three or more kids at home: Number of days read newspaper per week: 3, 2, 1, 7, 1, 0, 0, 0, 0, 2, 2 Use these raw data to construct a...
-
When someone rates a group at 10 on the ANES feeling thermometer, thats a pretty serious sign of dislike. I make the claim that the population of African American, female strong Democrats aged 17 to...
-
what the Central Limit Theorem is and why it is important

Study smarter with the SolutionInn App



