What volume of O2 at 760. mmHg and 23 C is required to synthesize 25.0 mol...
Fantastic news! We've Found the answer you've been seeking!
Question:
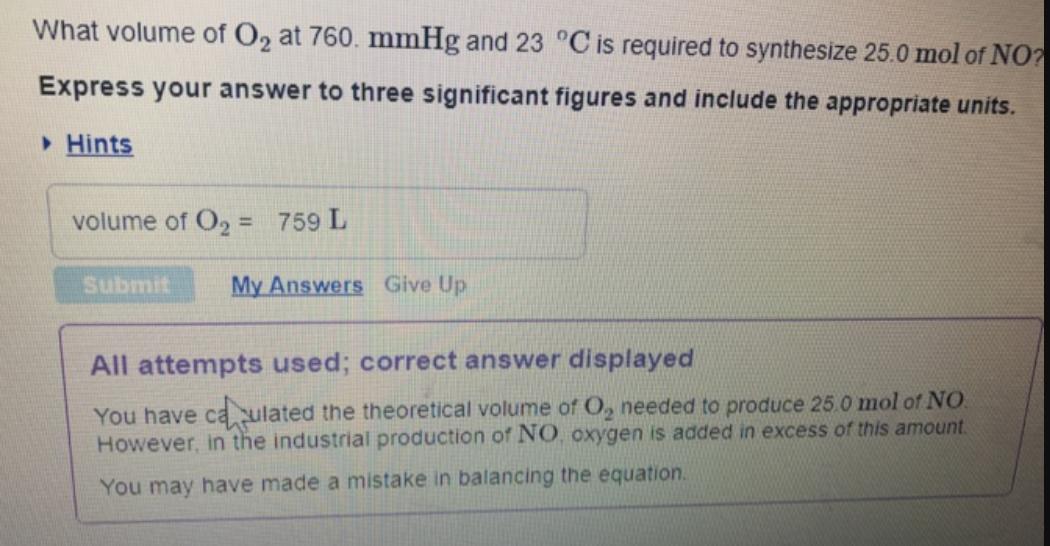
Transcribed Image Text:
What volume of O2 at 760. mmHg and 23 °C is required to synthesize 25.0 mol of NO? Express your answer to three significant figures and include the appropriate units. • Hints volume of O2 = 759 L %3D Submit My Answers Give Up All attempts used; correct answer displayed You have caulated the theoretical volume of O, needed to produce 25.0 mol of NO. However, in the industrial production of NO. oxygen is added in excess of this amount. You may have made a mistake in balancing the equation. What volume of O2 at 760. mmHg and 23 °C is required to synthesize 25.0 mol of NO? Express your answer to three significant figures and include the appropriate units. • Hints volume of O2 = 759 L %3D Submit My Answers Give Up All attempts used; correct answer displayed You have caulated the theoretical volume of O, needed to produce 25.0 mol of NO. However, in the industrial production of NO. oxygen is added in excess of this amount. You may have made a mistake in balancing the equation.
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
What volume of O2 is formed from the decomposition of 3.009 g of HgO if the gas has a pressure of 744 torr and a temperature of 122C?
-
A mole of gas at 0oC and 760 mmHg occupies 22.41 L. What is the volume at 20oC and 760 mmHg?
-
The man attempts to pull the log at C by using the three ropes. Determines the direction θ in which he should pull on his rope with a force P, so that he exerts a maximum force on the log. What...
-
Kamloops Company is a grocery wholesaler and is planning to expand its operations. The company has asked the bank for a loan to finance the expansion. Alphonzo, the companys manager, has prepared the...
-
The parameter values for a certain armature-controlled motor are KT = Kb = 0.05 N.m/A Ra = 0.8 La = 3x 10-3 H I = 8 x 10-5 kg.m2 Where I includes the inertia of the armature and that of the load...
-
Which method declarations can @SafeVarargs be correctly applied to? (Choose two.) A. protected void hum(double... tune) B. final int whistle(int length, float... measure) C. static void sing() D....
-
What happens to the probability that a particular person works when the wage rises? Does such a wage increase generate an income effect?
-
A summary of revenues and expenses for Norwalk Company for 2011 follows: Sales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $7,200,000 Cost of...
-
Deacon Company is a merchandising company that is preparing a budget for the three-month period ended June 30th. The following information is available Assets Deacon Company Balance Sheet March 31...
-
Lombard Ltd has been offered a contract for which there is available production capacity. The contract is for 20,000 identical items, manufactured by an intricate assembly operation, to be produced...
-
Perform a breadth-first search on the graph assuming that the vertices and adjacency lists are listed in alphabetical order. Show the breadth-first search tree that is generated. Consider the...
-
2. Consider the following figures. In each case decide whether the shaded portion in the figures represents the indicated fractions. Explain why or why not. 2 (a) (b) H 3. In each of the following...
-
16. (i) Show that the area of AABD: ACBD = |ADI: IDCI. (ii) ABCD below is a trapezium. Prove that Area c = Area d. (iii) Hence show that the area of the trapezium ABCD = Area a + Area b + 2 Area a....
-
We can reasonably approximate the coffee as simple water, with a molar mass of 18 and a density of 1 . How many moles of water are in your mug? 8 mol
-
You toss a tennis ball straight upward. At the moment it leaves your hand it is at a height of 1.5 m above the ground, and it is moving at a speed of 7.6 m/s. (a) How much time does it take for the...
-
1 1 Given an integer n 1, define J(n)=1+++ We want to extend the function J to other values than positive integers. The starting point is the equality n 1+t++ +t" = 1-th 1-t 1-1+1 1- -dt. Hint:...
-
2.A wheel of radius b rolls along the ground with constant forward speed Vo. Find the acceleration, relative to the ground, of any point on the rim. Vo 3 . P (Rotating coordinate fixed to a rolling...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
In the previous problem and the associated molecular model at the book's website, you considered the role of HOMOs and LUMOs in an SN2 reaction. (a) What is the LUMO in an SN1 reaction and in what...
-
Show how each of the following products could be synthesized from butanal: (a) 2-Ethyl-3-hydroxyhexanal (b) 2-Ethylhex-2-en-1-ol (c) 2-Ethylhexan-1-ol (d) 2-Ethylhexane-1, 3-diol (the insect...
-
Consider the following pairs of structures. Designate each chirality center as (R) or (S) and identify the relationship between them by describing them as representing enantiomers, diastereomers,...
-
Analyze the following solutions to the problem of Social Security. a. The retirement age is increased to 70. b. The FICA tax is increased. c. Income plus Social Security payments cannot exceed the...
-
Oregon proposed a solution to the health care costs problem that was widely criticized. Under this solution, the state paid only for common medical problems. Special and expensive problems would not...
-
Why is a third-party payer a problem? Private insurance companies are third-party payers, and yet they want to maximize profit. So wouldnt they ensure that the allocation of dollars was efficient?

Study smarter with the SolutionInn App


