9) How many asymmetric carbons are present in the compound below? Hc HC A) 0 B)...
Fantastic news! We've Found the answer you've been seeking!
Question:

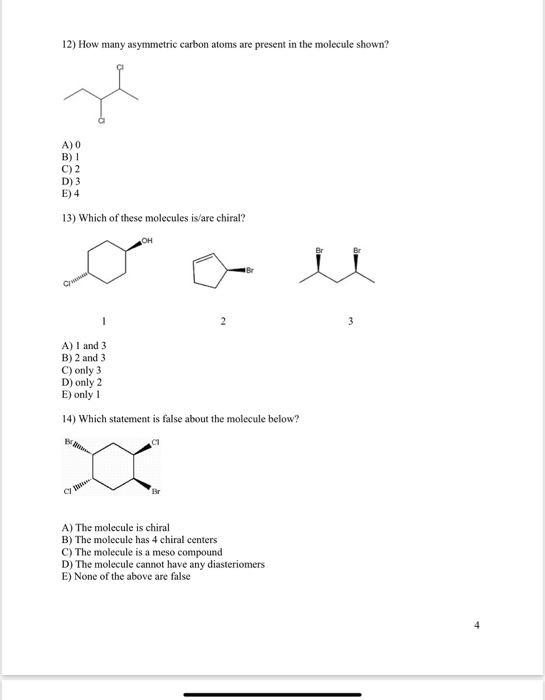
Transcribed Image Text:
9) How many asymmetric carbons are present in the compound below? H₂c H₂C A) 0 B) 1 C) 2 D) 3 E) 4 10) How many asymmetric carbons are present in the compound below? (H₂C)₂N A) 1 B) 2 C) 3 HO- HO A) 1 B) 2 C) 3 E) 5 D) 4 E) 5 11) How many asymmetric carbons are present in the compound below? CH₂OH .0 CH(CH₂)₂ OH OH OH CECCHS 12) How many asymmetric carbon atoms are present in the molecule shown? A) 0 B) 1 C) 2 D) 3 E) 4 13) Which of these molecules is/are chiral? A) 1 and 3 B) 2 and 3 OH Cy C) only 3 D) only 2 E) only 1 14) Which statement is false about the molecule below? X C1 2 Br A) The molecule is chiral B) The molecule has 4 chiral centers C) The molecule is a meso compound D) The molecule cannot have any diasteriomers E) None of the above are false 3 9) How many asymmetric carbons are present in the compound below? H₂c H₂C A) 0 B) 1 C) 2 D) 3 E) 4 10) How many asymmetric carbons are present in the compound below? (H₂C)₂N A) 1 B) 2 C) 3 HO- HO A) 1 B) 2 C) 3 E) 5 D) 4 E) 5 11) How many asymmetric carbons are present in the compound below? CH₂OH .0 CH(CH₂)₂ OH OH OH CECCHS 12) How many asymmetric carbon atoms are present in the molecule shown? A) 0 B) 1 C) 2 D) 3 E) 4 13) Which of these molecules is/are chiral? A) 1 and 3 B) 2 and 3 OH Cy C) only 3 D) only 2 E) only 1 14) Which statement is false about the molecule below? X C1 2 Br A) The molecule is chiral B) The molecule has 4 chiral centers C) The molecule is a meso compound D) The molecule cannot have any diasteriomers E) None of the above are false 3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of these molecules is unsaturated? [Section 24.3] CH2 CH2 CH2 CH3CH2CH2CH3 CH2 CH2 CHC-OH CH CHCH, CH3CH CHCH3
-
Which of these molecules most readily undergoes an addition reaction? [Section 24.3] 00 CHCHO-OH CH CH CH CHC OH CH2CH2 NH2
-
How many unpaired electrons are present in the tetrahedral ion FeCl 4 - ?
-
In your opinion, was Saks' zero tolerance policy for employee theft reasonable? Was the policy likely cost-effective? Defend your answers.
-
Why would a company choose to borrow money rather than issue additional stock?
-
Kellogg Company produces, markets, and distributes cereal and food products including CheezIt, Coco Pops, Rice Krispies, and Pringles. The following partial income statements (in millions) were...
-
Steven Sanchez worked as a warehouse employee for Gruma Corporation. In December 2016, Sanchez was presented an arbitration agreement during a meeting with George Tate, the human resources manager...
-
Patel, CPA, has completed the audit of the financial statements of Bellamy Corporation as of and for the year ended December 31, 2011. Patel also audited and reported on the Bellamy financial...
-
(d) What is the value of "j" after the execution of the partial code in Figure 4.2? Explain your answer. int j = 10; for (int i = 0; 0; i
-
Bob Burley and his brother Buford ran the best restaurant in Dallas, Texas. Many out-of-towners would visit Dallas and go to Burleys Biscuits, Beef, and Veggies for a good wholesome meal. One thing...
-
Write a paper on Jesus in the Gospel of Mark, answering the following questions: When does his ministry begin? What traits do you notice? How does Jesus confront the domination system? How would you...
-
1. Design one class that represents these types of objects: CINEMA PRESENTS FILM TITLE SCREEN ADULT standard 14 PRICE 8.60 DATE PROGRAMME TIME 06/11/2011 18:00 RATING 12A PRINTABLE MOVIE NIGHT...
-
If logsy-A=Blogsx, where x>0 and y > 0, then y is equal to Oa. x85A Ob. b. x8 5A O c. x-A Od. y B(5A)
-
3. Given f(x)=2x-12x+18x a) Use the second derivative sign chart to determine the intervals on which f(x) is concave up and concave down. b) Give the coordinates of any inflection points.
-
Solve the given differential equation. y= y'-y=13 (Use C as the arbitrary constant.) ID: 31.2.17
-
John raises Angus cattle and sells them to a packing house. McDonald's then sells the meat supplied by the packer as hamburgers. In this scenario, which of the following economic terms best describes...
-
At GameStudios Inc., the output of a specific process is valued at 112 per unit. The accounting department provided the following information about the process for the past 5 weeks: Week 1 Week 2...
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
Determine whether each of the following compounds is a cis isomer or a trans isomer a. b. c. d. e. f. Cl Br CH3 CH3 Br Br CH3 Cl CH3 CH3 CH
-
How could you prepare the following compounds using a starting material containing no more than three carbon atoms? a. b. CH3 CH CH CHCHCH O Br CH CH2CH2CCH3
-
What ketones would be formed from the acid-catalyzed hydration of 3-heptyne?
-
What is a business strategy? Do you agree with the definition proposed? Illustrate your answer with examples.
-
Consider one of the following firms. Read the description of a business strategy in the text. Go to the firm's website and use it to gain an understanding of the business strategy. Look at elements...
-
Which quote at the front of the chapter do you find the most insightful? Why? Under what circumstances would its implications not hold?

Study smarter with the SolutionInn App



