Calculate the equilibrium constant K p for the reaction at a temperature of298 K. + Gibbs Free
Fantastic news! We've Found the answer you've been seeking!
Question:
Calculate the equilibrium constant Kp for the reaction at a temperature of298 K.
Transcribed Image Text:
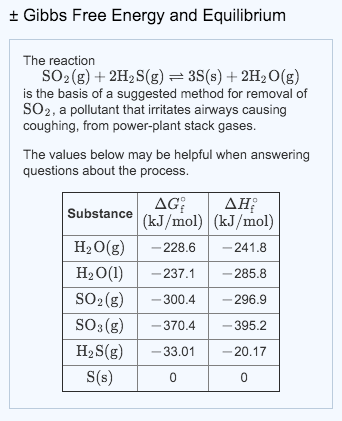
+ Gibbs Free Energy and Equilibrium The reaction SO₂(g) + 2H₂S(g) = 3S(s) + 2H₂O(g) is the basis of a suggested method for removal of SO₂, a pollutant that irritates airways causing coughing, from power-plant stack gases. The values below may be helpful when answering questions about the process. AG ΔΗ Substance (kJ/mol) (kJ/mol) H₂O(g) -228.6 -241.8 H₂O(1) -237.1 -285.8 SO₂ (g) -300.4 -296.9 SO3(g) -370.4 -395.2 H₂S(g) - 33.01 -20.17 S(s) 0 0 + Gibbs Free Energy and Equilibrium The reaction SO₂(g) + 2H₂S(g) = 3S(s) + 2H₂O(g) is the basis of a suggested method for removal of SO₂, a pollutant that irritates airways causing coughing, from power-plant stack gases. The values below may be helpful when answering questions about the process. AG ΔΗ Substance (kJ/mol) (kJ/mol) H₂O(g) -228.6 -241.8 H₂O(1) -237.1 -285.8 SO₂ (g) -300.4 -296.9 SO3(g) -370.4 -395.2 H₂S(g) - 33.01 -20.17 S(s) 0 0
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
You obtain kinetic data for a reaction at a set of different temperatures. You plot ln k versus 1/T and obtain the following graph: Suggest a molecular-level interpretation of these unusual data. 1/T
-
Calculate the KP for this reaction at 298 K if the Keq = 1.76 103. 3O2 (g) + 2O3(g)
-
Calculate the KP for this reaction at 310 K if the Keq = 6.22 103. 4NO2 (g) + O2 (g) + 2N2O5 (g)
-
Successful interviews are results of completing pre-interview tasks, such as Identifying the interviewee, doing research on the topic and interviewee, choosing a location or method for the interview,...
-
Quattro Technologies, a hydraulic manufacturer in the aeronautics industry, has reported steadily increasing earnings over the past few years. The company reported net income of $120 million in 2013...
-
At the end of the current year, $47,700 of fees have been earned but not billed to clients. a. What is the adjustment to record the accrued fees? Indicate each account affected, whether the account...
-
Larry Riley formed a rental agreement for a self-storage unit with Iron Gate Self Storage in December 2003. The agreement stated that a maximum of \($5,000\) of personal property could be stored in...
-
During the first month of operations ended July 31, 2014, YoSan Inc. manufactured 2,400 flat panel televisions, of which 2,000 were sold. Operating data for the month are summarized as follows:...
-
as Suppose X is a normal variable with mean and variance 2. Let f(x) be the density function of X. For a constant 0, define the tilted density function fo(x) = f(x)ex Mo where Me is the normalization...
-
In 20X2, the new CEO of Watsontown Electric Supply became concerned about the companys apparently deteriorating financial position. Wishing to make certain that the grim monthly reports he was...
-
Discuss the key threats to maritime security, including but not limited to piracy, terrorism, human trafficking, drug trafficking, and illegal fishing. Explain the impact of these threats on global...
-
Express in polar form the product of the given complex numbers. 5(cos 125+i sin 125). 4(cos 31+i sin 31) 20(cos 156+i sin 156) 20(cos 94 +i sin 94) 20(cos 120+i sin 120) 20(cos 90 +i sin 90)
-
Which of these shows a pair of functions that are BOTH undefined at an angle of = 3T ? 2 tan a and cota csc a and co't a Osin a and cos a O sec a and tan a
-
10. A company estimates that it will sell N(x) units of a product after spending $x thousand on advertising. N(x)=4x-0.25x +500 0x12 a) How much money should the company spend on advertising to reach...
-
1 pts Calculate the refining margin for crude oil X in $ per barrel. Type the margin into the blank to two decimal places. Do not type the $ sign. So if you calculate the answer to be 5.30, type 5.30...
-
12. 12 points Using the iterative method (forward or backward substitution) solve the following recurrence relations with the given initial conditions. Show the steps of your calculation. Note: you...
-
In the formula, the weights of autonomy and feedback are how many times the weight of variety, identity, and significance? SPS= SPS= SPS= V1+V2+11+12+S1+S2 6 6 X X X A1+A2 2 2 X X X 11 F1+F2 2 2
-
The swap spread is the difference between the swap rate and the equivalent-maturity Treasury bond yield. Explain why a widening swap spread may be a signal of deteriorating economic conditions. Plot...
-
Consider the reaction Would you expect this reaction to be endothermic or exothermic? Why? N2O4 is a colorless gas; NO2 is red-brown. Would you expect a mixture of these gases to become more or less...
-
What are some characteristics of the adhesive used for Post-it Notes?
-
The following is a table of some of the oxoacids of the halogens. Which acid in the table is the strongest? Which is the weakest? Explain how you arrived at your answers. Chlorine Bromine Iodine HOBr...
-
The following are the financial statements for Nailsea plc for the years ended 30 June 2020 and 2021 There were no disposals of non-current assets in either year. Dividends were paid in 2020 and 2021...
-
The statements of financial position of Axis plc as at 31 December 2020 and 2021 and the income statement for the year ended 31 December 2021 were as follows: 1 During the year, plant (a non-current...
-
Consider the following: 1 A motor car manufacturer offers a three-year warranty on all cars that it produces. The warranty undertakes to make good any defects arising from the manufacturing process....

Study smarter with the SolutionInn App



