9B.1: You are trying to create a new alloy made of recently discovered elements Virginium (Vr)...
Fantastic news! We've Found the answer you've been seeking!
Question:

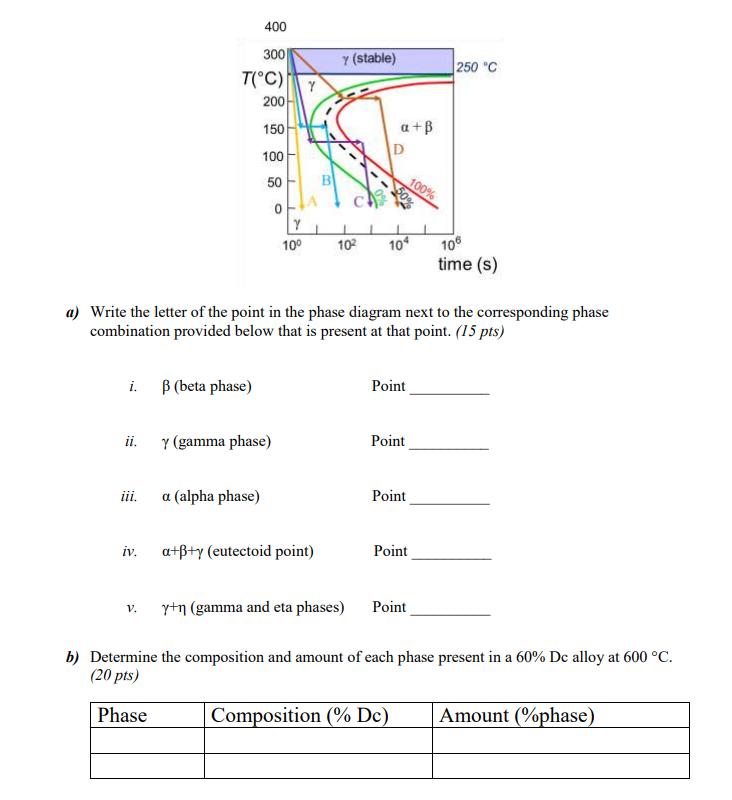

Transcribed Image Text:
9B.1: You are trying to create a new alloy made of recently discovered elements Virginium (Vr) and Washingtonium (Dc). A fellow scientist created the phase diagram for the whole system and a TTT diagram for a system of 60% Vr, 40% Dc starting at 400 C, but needs help to answer the following questions. Temperature, C 1600- 15383 1475 1400- 1394 8 (Vr) 1200- 1000- 912 800- 600- 400- y+a 200- Vr L 250 a+ Y+B 690 & (Dc) 1230 1246 1155 1138 1100 n (De) 10 20 30 40 50 60 80 70 80 90 100 wt.% Dc Bo 727 400 300 T(C) Y (stable) 250 C Y 200 150 D a+ 50 0 680 100 B 100% 10 102 10 106 time (s) a) Write the letter of the point in the phase diagram next to the corresponding phase combination provided below that is present at that point. (15 pts) i. (beta phase) Point ii. y (gamma phase) Point iii. a (alpha phase) Point iv. a++y (eutectoid point) Point V. y+n (gamma and eta phases) Point b) Determine the composition and amount of each phase present in a 60% Dc alloy at 600 C. (20 pts) Phase Composition (% Dc) Amount (%phase) c) You heat the system and hold it at point B, then slowly cool it at constant composition to room temperature. Which of the following is present when the process is complete (circle the number of all that apply) (12 pts) HINT: Point B is directly above Point C. Pro-eutectoid y phase 1. 2. Pro-eutectoid a phase 3. Pro-eutectoid phase 4. Eutectoid y gamma phase 5. Eutectoid a phase 6. Eutectoid phase d) Use the phase diagram and/or TTT diagram for the 60% Vr, 40% Dc alloy to determine the following (8 pts): i. The steps required to process the alloy such that you end up with 50% retained y in the microstructure at 0 C. (Circle One) ii. Process A Process B Process C Process C What type of phase is present after reaching room temperature following Process A? (Circle One) Pro-eutectoid Hypereutectoid Precipitate Metastable 9B.1: You are trying to create a new alloy made of recently discovered elements Virginium (Vr) and Washingtonium (Dc). A fellow scientist created the phase diagram for the whole system and a TTT diagram for a system of 60% Vr, 40% Dc starting at 400 C, but needs help to answer the following questions. Temperature, C 1600- 15383 1475 1400- 1394 8 (Vr) 1200- 1000- 912 800- 600- 400- y+a 200- Vr L 250 a+ Y+B 690 & (Dc) 1230 1246 1155 1138 1100 n (De) 10 20 30 40 50 60 80 70 80 90 100 wt.% Dc Bo 727 400 300 T(C) Y (stable) 250 C Y 200 150 D a+ 50 0 680 100 B 100% 10 102 10 106 time (s) a) Write the letter of the point in the phase diagram next to the corresponding phase combination provided below that is present at that point. (15 pts) i. (beta phase) Point ii. y (gamma phase) Point iii. a (alpha phase) Point iv. a++y (eutectoid point) Point V. y+n (gamma and eta phases) Point b) Determine the composition and amount of each phase present in a 60% Dc alloy at 600 C. (20 pts) Phase Composition (% Dc) Amount (%phase) c) You heat the system and hold it at point B, then slowly cool it at constant composition to room temperature. Which of the following is present when the process is complete (circle the number of all that apply) (12 pts) HINT: Point B is directly above Point C. Pro-eutectoid y phase 1. 2. Pro-eutectoid a phase 3. Pro-eutectoid phase 4. Eutectoid y gamma phase 5. Eutectoid a phase 6. Eutectoid phase d) Use the phase diagram and/or TTT diagram for the 60% Vr, 40% Dc alloy to determine the following (8 pts): i. The steps required to process the alloy such that you end up with 50% retained y in the microstructure at 0 C. (Circle One) ii. Process A Process B Process C Process C What type of phase is present after reaching room temperature following Process A? (Circle One) Pro-eutectoid Hypereutectoid Precipitate Metastable
Expert Answer:

Related Book For 

International Marketing And Export Management
ISBN: 9781292016924
8th Edition
Authors: Gerald Albaum , Alexander Josiassen , Edwin Duerr
Posted Date:
Students also viewed these chemical engineering questions
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
Joseph Kent started business on 1 July 2018 as a joiner making conservatories. His taxadjusted profits (before deduction of capital allowances) were as follows: Private use of both cars has been...
-
a. What are some types of real options? b. What are five possible procedures for analyzing a real option? c. Tropical Sweets is considering a project that will cost $70 million and will generate...
-
Graph the system of inequalities. Sx + y? < 9 + y 2 3
-
Consider the Gallo strategic decision. Describe how you would go about evaluating that decision.
-
I know headquarters wants us to add that new product line, said Fred Halloway, manager of Kirsi Products East Division. But I want to see the numbers before I make a move. Our divisions return on...
-
1. Create a demand graph using the following table of values: PRICE QUANTITY 500 10 20 450 30 400 40 350 50 300 60 250 70 200 2. Create a supply graph using the following table of values: PRICE...
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study long-term debt...
-
1. The Catawba River provides water for communities in two states, North and South Carolina. Suppose that 110 (MAF) of water from the river are available for use. Marginal willingness to pay for...
-
Pedregon Corporation has provided the following information: Direct materials Direct labor Variable manufacturing overhead Fixed manufacturing overhead Sales commissions Variable administrative...
-
Statement of stockholders' equity Financial information related to Webber Company for the month ended June 30, 20Y7, is as follows: Common Stock, June 1, 20Y7 Stock issued in June Net income for June...
-
In preparing Culver Inc.'s statement of cash flows for the year ended December 31, 2025, the following amounts were available: Collection of note receivable - $623000 Issuance of bonds payable -...
-
Current Attempt in Progress Flounder Company provided the following information on selected transactions during 2024: Dividends paid to preferred stockholders Loans made to affiliated corporations...
-
At Sandhill Ltd., prepaid costs are debited to expense when cash is paid and unearned revenues are credited to revenue when the cash is received. During January of the current year, the following...
-
[o-algebra : Properties and Construction]. (a) Show that a o-algebra is also an algebra. (b) Given a sample space N and a o-algebra F of the subsets of 2, show that if A, B E F, A\B and AAB, the...
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Discuss what you consider to be the ideal approach for establishing an export transfer price.
-
What is e-business?
-
Licensing seems to be a fairly safe way for a manufacturer to produce in a foreign market for the first time. Comment.
-
Bullying needs to be able to be defined in behavioral terms so that a workplace bullying policy can move from a concept to an operational reality. If bullying could be behaviorally defined, the...
-
The beryllium sensitization of OSHAs compliance officers has raised concerns for the thousands of industry workers who are exposed to beryllium. OSHA officers visit the workplaces only sporadically,...
-
OSHA has been criticized for its inadequate beryllium exposure standards. The agency did try to push through lower exposure limits in the l970s, but companies stopped passage of the new standards by...

Study smarter with the SolutionInn App


