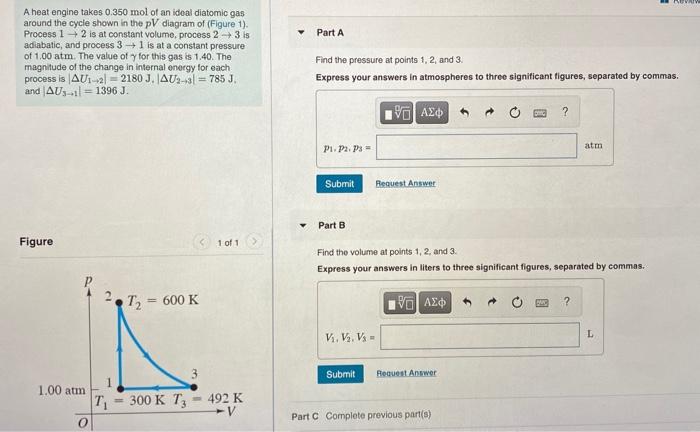
A heat engine takes 0.350 mol of an ideal diatomic gas around the cycle shown in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A heat engine takes 0.350 mol of an ideal diatomic gas around the cycle shown in the pV diagram of (Figure 1). Process 1 2 is at constant volume, process 2 +3 is adiabatic, and process 31 is at a constant pressure of 1.00 atm. The value of y for this gas is 1.40. The magnitude of the change in internal energy for each process is AU1-al = 2180 J. JAU:-al = 785 J. and AUs1l = 1396 J. Part A Find the pressure at points 1, 2, and 3. Express your answers in atmospheres to three significant figures, separated by commas. atm P1. Pa. Ps = Submit Request Answer Part B Figure 1 of 1 Find the volume at points 1, 2, and 3. Express your answers in liters to three significant figures, separated by commas. T2 = 600 K L. V, V, Và = Submit Request Answer 1.00 atm T 492 K -V 300 K T3 %3D Part C Complete previous part(s) Revidwi Constants You decide to use your body as a Carnot heat engine. The operating gas is in a tube with one end in your mouth (where the temperature is 37.0 C) and the other end at the surface of your skin, at 30.0C. Part A What is the maximum etficiency of such a heat engine? Express your answer in percents. ? Submit Reguest Angwer Part B Suppose you want to use this human engine to ift a 3.00 kg box from the foor to a tabletop 1.20 m above the floor. How much must you increase the gravitational potential energy? Express your answer with the approprinte units. U- Value Units Submit leguest Anawer You decide to use your body as a Camot heat engine. The operating gas is in a tube with one end in your mouth (where the temperature is 37.0 C) and the other end at the surface of your skin, at 30.0 C I Review I Constants Part C How much heat input is needed to accompish the in? Express your answer with the appropriate units. HA Q= Value Units Submit Beguest Answer Part D How many 350 calorie (those are food calories, remember) candy bars must you eat to t the box in this way? Recall that 80% of the food energy goes into heat. Express your answer using two significant figures. N= candy bars A heat engine takes 0.350 mol of an ideal diatomic gas around the cycle shown in the pV diagram of (Figure 1). Process 1 2 is at constant volume, process 2 +3 is adiabatic, and process 31 is at a constant pressure of 1.00 atm. The value of y for this gas is 1.40. The magnitude of the change in internal energy for each process is AU1-al = 2180 J. JAU:-al = 785 J. and AUs1l = 1396 J. Part A Find the pressure at points 1, 2, and 3. Express your answers in atmospheres to three significant figures, separated by commas. atm P1. Pa. Ps = Submit Request Answer Part B Figure 1 of 1 Find the volume at points 1, 2, and 3. Express your answers in liters to three significant figures, separated by commas. T2 = 600 K L. V, V, Và = Submit Request Answer 1.00 atm T 492 K -V 300 K T3 %3D Part C Complete previous part(s) Revidwi Constants You decide to use your body as a Carnot heat engine. The operating gas is in a tube with one end in your mouth (where the temperature is 37.0 C) and the other end at the surface of your skin, at 30.0C. Part A What is the maximum etficiency of such a heat engine? Express your answer in percents. ? Submit Reguest Angwer Part B Suppose you want to use this human engine to ift a 3.00 kg box from the foor to a tabletop 1.20 m above the floor. How much must you increase the gravitational potential energy? Express your answer with the approprinte units. U- Value Units Submit leguest Anawer You decide to use your body as a Camot heat engine. The operating gas is in a tube with one end in your mouth (where the temperature is 37.0 C) and the other end at the surface of your skin, at 30.0 C I Review I Constants Part C How much heat input is needed to accompish the in? Express your answer with the appropriate units. HA Q= Value Units Submit Beguest Answer Part D How many 350 calorie (those are food calories, remember) candy bars must you eat to t the box in this way? Recall that 80% of the food energy goes into heat. Express your answer using two significant figures. N= candy bars
Expert Answer:

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these physics questions
-
The temperature of 3.00 mol of an ideal diatomic gas is increased by 40.0 Co without the pressure of the gas changing. The molecules in the gas rotate but do not oscillate. (a) How much energy is...
-
A monatomic ideal gas is taken around the cycle shown in Fig. 20.29 in the direction shown in the figure. The path for process c → a is a straight line in the pV-diagram. (a) Calculate Q, W, and...
-
A reversible compression of 1 mol of an ideal gas in a piston/cylinder device results in a pressure increase from 1 bar to P2 and a temperature increase from 400 K to 950 K. The path followed by the...
-
An increase in the supply of U.S. dollars by the Federal Reserve will raise the value of the dollar because it will stimulate U.S. economic growth raise the value of the dollar because it will lead...
-
Consider again the conditions of Exercise 7. (a) If it is observed that X = 1/2, what predicted value of Y will have the smallest M.S.E.? (b) What will be the value of this M.S.E.?
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this book. The following selected information (in U.S. thousands) has been taken from these,...
-
Two springs are hooked together end to end. When a \(4.0-\mathrm{kg}\) brick is suspended from one end of the combination, the combination stretches \(0.15 \mathrm{~m}\) beyond its relaxed length....
-
The heat capacity at constant pressure of hydrogen cyanide is given by the expression Cp[J/(mol C)] = 35.3 + 0.0291T(C) (a) Write an expression for the heat capacity at constant volume for HCN,...
-
Your firm has total debt of $4, 180 and debt equity ratio 0.79. What is the amount of total assets? Round your answer to the nearest number, no percentage sign. Answer
-
1. Relate the material in this case to the concept of the total retail experience. 2. What uncontrollable factors will affect a retailers ability to undertake the suggestions cited in the case? How...
-
IVANA Ltd commenced business on 1st January 2024 making one product only, which sells for 160 per item. The production and sales data for each of the first three months of 2024 were as follows: Sales...
-
What is the current yield for a bond with a current price of $1,200, a face value of $1,600, and a coupon rate of 5%? Round your answer to the nearest tenth.
-
1. Mark decides to open a lemonade stand. To start, she needs $50 for a licence, $30 for materials (such as cups, lemons, sugar, ice), and $20 for a stand. He expects to sell 100 cups of lemonade at...
-
Tax planning for individuals and families is an ongoing process intended to reduce the overall taxes owed by the family and usually involves experiments with different tax-planning strategies....
-
Find each of the following products. 2 (a) 13 (b) 2 -2 403 41
-
Effective accounting practices are essential for businesses to achieve financial stability, optimize resource allocation, and make informed decisions for long-term success. In the dynamic and...
-
Use the following returns for X and Y. Year 12345 Returns C. X 21.5% -16.5 9.5 19.0 4.5 a. Average return b. Variance Y 25.5% 3.5 27.5 - 14.0 31.5 a. Calculate the average returns for X and Y. (Do...
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
A sample of gas expands from V1 = 1.0 m3 and P1 = 40 Pa to V2 = 4.0 m3 and P2 = 10 Pa along path B in the p-V diagram in Figure. It is then compressed back to V1 along either path A or path C....
-
The (United States) National Electric Code, which sets maximum safe currents for insulated copper wires of various diameters, is given (in part) in the table. Plot the safe current density as a...
-
A damped harmonic oscillator consists of a block (m = 2.00 kg), a spring (k = 10.0 N/m), and a damping force (F = bv). Initially, it oscillates with an amplitude of 25.0 cm; because of the damping,...
-
Suppose, for Problem 13.20, that your PV was 70 and your EV was 95. Recalculate the SPI and estimated time to completion for the project with this new data. Problem 13.20 You are calculating the...
-
You are calculating the estimated time to completion for a project of 12 months duration and a budgeted cost of $500,000. Assuming the following information, calculate the Schedule Performance Index...
-
Go to www.nu-solutions.com/downloads/earned_ value_lite.pdf and access the article by Q. W. Fleming and J. M. Koppelman, Earned Value Lite: Earned Value for the Masses. From your reading, summarize...

Study smarter with the SolutionInn App


