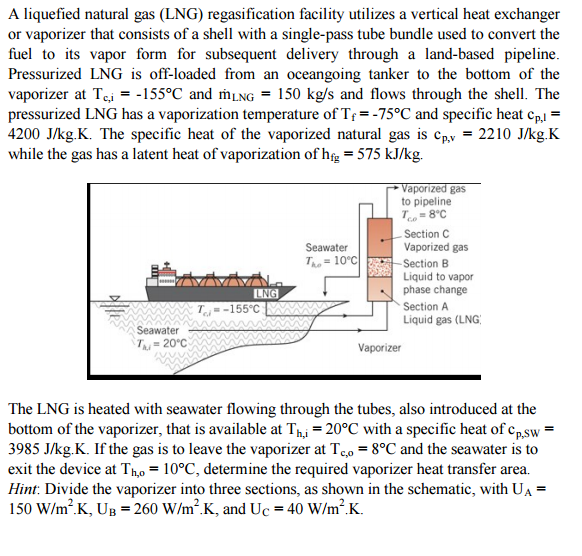
? A liquefied natural gas (LNG) regasification facility utilizes a vertical heat exchanger or vaporizer that consists
Fantastic news! We've Found the answer you've been seeking!
Question:
? 
Transcribed Image Text:
A liquefied natural gas (LNG) regasification facility utilizes a vertical heat exchanger or vaporizer that consists of a shell with a single-pass tube bundle used to convert the fuel to its vapor form for subsequent delivery through a land-based pipeline. Pressurized LNG is off-loaded from an oceangoing tanker to the bottom of the vaporizer at Tc,i = -155°C and mLNG = 150 kg/s and flows through the shell. The pressurized LNG has a vaporization temperature of T = -75°C and specific heat Cp,l = 4200 J/kg.K. The specific heat of the vaporized natural gas is Cp,v = 2210 J/kg.K while the gas has a latent heat of vaporization of hfg = 575 kJ/kg. Seawater Thi = 20°C LNG T-155°C Seawater = 10°C The Vaporized gas to pipeline To=8°C Section C Vaporized gas -Section B Liquid to vapor phase change Vaporizer Section A Liquid gas (LNG) The LNG is heated with seawater flowing through the tubes, also introduced at the bottom of the vaporizer, that is available at Th,i = 20°C with a specific heat of Cp,sw= 3985 J/kg.K. If the gas is to leave the vaporizer at To = 8°C and the seawater is to exit the device at Tho = 10°C, determine the required vaporizer heat transfer area. Hint: Divide the vaporizer into three sections, as shown in the schematic, with UA = 150 W/m².K, UB=260 W/m².K, and Uc = 40 W/m².K. A liquefied natural gas (LNG) regasification facility utilizes a vertical heat exchanger or vaporizer that consists of a shell with a single-pass tube bundle used to convert the fuel to its vapor form for subsequent delivery through a land-based pipeline. Pressurized LNG is off-loaded from an oceangoing tanker to the bottom of the vaporizer at Tc,i = -155°C and mLNG = 150 kg/s and flows through the shell. The pressurized LNG has a vaporization temperature of T = -75°C and specific heat Cp,l = 4200 J/kg.K. The specific heat of the vaporized natural gas is Cp,v = 2210 J/kg.K while the gas has a latent heat of vaporization of hfg = 575 kJ/kg. Seawater Thi = 20°C LNG T-155°C Seawater = 10°C The Vaporized gas to pipeline To=8°C Section C Vaporized gas -Section B Liquid to vapor phase change Vaporizer Section A Liquid gas (LNG) The LNG is heated with seawater flowing through the tubes, also introduced at the bottom of the vaporizer, that is available at Th,i = 20°C with a specific heat of Cp,sw= 3985 J/kg.K. If the gas is to leave the vaporizer at To = 8°C and the seawater is to exit the device at Tho = 10°C, determine the required vaporizer heat transfer area. Hint: Divide the vaporizer into three sections, as shown in the schematic, with UA = 150 W/m².K, UB=260 W/m².K, and Uc = 40 W/m².K.
Expert Answer:

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these mechanical engineering questions
-
Show reactions that could be used to convert the epoxide to these compounds. More than one step may benecessary. SCH3 OH b) CH,O- a) Br
-
Which method of translation is used to convert the financial statements when a foreign subsidiary operates in a highly inflationary economy?
-
Which functions can be used to convert the letter case of character values?
-
You work as a manager in the admission department of company. You were asked by your department head to present and analyze the qualifying test scores of 40 employees who are applying for 5-year...
-
The financial statements of Amazon.com, Inc. are presented in Appendix D. Financial statements of Wal-Mart Stores, Inc. are presented in Appendix E. Instructions (a) Based on the information in the...
-
In 2013, the average earnings of a high school graduate were $30,286. At $50,738, the average earnings of a recipient of a bachelor's degree were about 66% higher. (a) Construct a misleading graph...
-
Refer to information in QS 21-14. Compute the overhead volume variance for November and classify it as favorable or unfavorable. Data From QS 21-14 AirPro Corp. reports the following for November....
-
Bracewell Company reported net income of $195,000 for 2014. Bracewell also reported depreciation expense of $40,000 and a gain of $5,000 on disposal of plant assets. The comparative balance sheet...
-
8. Two dice are rolled. Let A represent rolling a sum greater than 7. Let B represent rolling a sum that is a multiple of 3. Determine n(AB). A. 5 B. 8 C. 12 D. 15 Long Answer: Show all work for the...
-
A traffic signal has a 60-second cycle length. For the travel direction of interest, the effective red and green times are 30 seconds each, the arrival rate is constant at 20 vehs/min and the...
-
Jonas Materials Science (JMS) purchases its materials from several countries. As part of its cost-control program, JMS uses a standard cost system for all aspects of its operations, including...
-
E. Go over the Araling Panlipunan Curriculum Guide. Identify a competency where you can use moral dilemma as a teaching technique. Formulate an example of a moral dilemma which you can use to teach...
-
Explain the observed difference in median hourly pay rates between customer service representatives and roofers. Your answer should be based on the information below and your understanding of...
-
Informative Speech Self-Evaluation help an evaluation of your Informative Speech, covering each of these items: 1. Preparation for the speech 2. Practicing for the speech 3. Main Points 4....
-
1. [12] Let a1, a2,..., an be a sequence of real numbers, for some n 1. A SUM-BOX is an ADT that stores the sequence and supports the following operations (S is a given SUM-BOX): PARTIAL-SUM(S, m):...
-
Hello there, can you do the below questions? 1) Select four poems to discuss from the ones that are listed in the overview. 2) Do the following for each poem: Give a brief summary of the poem and...
-
1. What was your first job? My first job was milking cows for my uncle. He was a dairy farmer in Ireland and it was kind of a holiday job. My first real paying job just after I graduated was working...
-
In a nonmagnetic medium, E = 50 cos (10 9 t 8x) a y + 40 sin (10 9 t 8x) a z V/m find the dielectric constant r and the corresponding H.
-
A process for the production of 1, 3-butadiene results from the catalytic dehydrogenation at atmospheric pressure of 1-butene according u> the reaction: C4H8(g) ( C4H6(g) + H2(g) To suppress side...
-
A steam power plant operates on the cycle of Fig. 8,4, For one of the following sets of operating conditions, determine the steam rate, the heal-transfer rates in the boiler and condenser, and the...
-
Natural gases arc rarely pure methane; they usually also contain other light hydrocarbons and nitrogen. Determine on expression for the standard heat of combustion as a function of composition for a...
-
We are a global, science-led biopharmaceutical business. Return to shareholders Revenue from the sale of our medicines generates cash flow, which helps us fund business investment. It also enables us...
-
Our mission and supporting strategies The FRCs mission is to promote high quality corporate governance and reporting to foster investment. Why have we landed on this mission? The capital markets are...
-
National Express and Stagecoach are both major providers of transport services in the UK. Here we compare their accounts of financial performance in their UK Bus divisions. National Express plc UK...

Study smarter with the SolutionInn App


