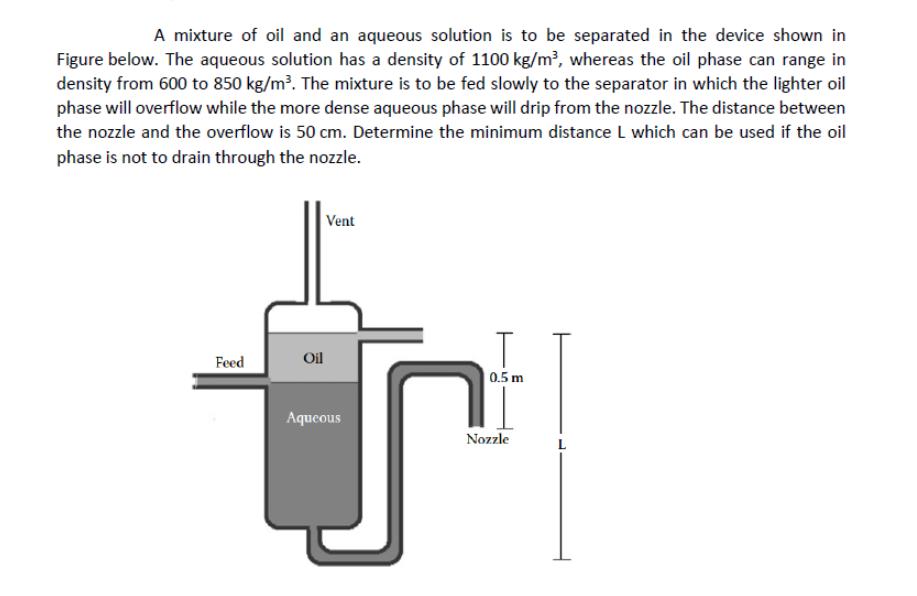
A mixture of oil and an aqueous solution is to be separated in the device shown...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A mixture of oil and an aqueous solution is to be separated in the device shown in Figure below. The aqueous solution has a density of 1100 kg/m³, whereas the oil phase can range in density from 600 to 850 kg/m³. The mixture is to be fed slowly to the separator in which the lighter oil phase will overflow while the more dense aqueous phase will drip from the nozzle. The distance between the nozzle and the overflow is 50 cm. Determine the minimum distance L which can be used if the oil phase is not to drain through the nozzle. Feed Vent Oil Aqueous 0.5 m Nozzle A mixture of oil and an aqueous solution is to be separated in the device shown in Figure below. The aqueous solution has a density of 1100 kg/m³, whereas the oil phase can range in density from 600 to 850 kg/m³. The mixture is to be fed slowly to the separator in which the lighter oil phase will overflow while the more dense aqueous phase will drip from the nozzle. The distance between the nozzle and the overflow is 50 cm. Determine the minimum distance L which can be used if the oil phase is not to drain through the nozzle. Feed Vent Oil Aqueous 0.5 m Nozzle
Expert Answer:
Answer rating: 100% (QA)
Final answer Approximate 0647m Detailed Explanation To prevent the oil phase from draining through t... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Journalize the 10 transactions for your company using excel (must include descriptions). Your opening entry should include the following 4 items: Bank $10,000 Supplies $5,000 Bank Loan $20,000...
-
A misture containing 500 wt% acetone and 50.0 wt% water is to be separated into two streams?one enriched in acetone. the other in water. The separation process consists of extraction of the acetone...
-
If the Federal Government increases taxes:What will be the effect on money demand, money supply, and interest rates? Money demand decreases, money supply is unchanged, and interest rates decrease...
-
Why do purchases appear as expenses on an income statement?
-
The Palm Garden Greenhouse specializes in raising carnations that are sold to florists. Carnations are sold for $3.00 per dozen; the cost of growing the carnations and distributing them to the...
-
Describe various forms of child abuse, how to recognize it, and reporting requirements.
-
Carolinas Golf School, Inc., completed the following transactions during October, 2012: Oct 1 Prepaid insurance for October through December, $900. 4 Performed services (gave golf lessons) on...
-
Required Information [The following Information applies to the questions displayed below.] Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net...
-
Bottle-Up, Inc., was organized on January 8, 2010, and made its S election on January 24, 2010. The necessary consents to the election were filed in a timely manner. Its address is 1234 Hill Street,...
-
1 3 7 1 point The intentional removal of life-support equipment can be considered the crime of I_____ battery assault murder 0000 petty theft
-
What kind of training should companies be offering to recruit and develop local management talent, particularly in emerging markets?
-
One reason for expatriate assignment failurean employees premature return from an international assignmentis culture shock. What is culture shock? Provide some examples of culture shock contributing...
-
How does a global management team help a company to coordinate host country and cross-border business?
-
Many MNCs choose divergent labor practices, rather than convergent, and adapt their HR policies to local conditions. Find examples of companies that have been accused of running sweatshops and...
-
In addition to financial compensation, what other variables can play a role in compensation that an IHRM can leverage in emerging markets? Provide examples from different cultures.
-
Sketch the resulting DSB-SC AM signal from the oscilloscope. Determine values of the total power, Pr, and side band power, PSB- What is the maximum value of the DSB-SC AM signal efficiency? Find the...
-
r = 0.18 Find the coefficients of determination and non-determination and explain the meaning of each.
-
Have each group member explain to the group what happens if a system at equilibrium is subject to one of the following changes and why: a. The concentration of a reactant is increased b. A solid...
-
What is the mass, in grams, of each elemental sample? a. 2.3 * 10 -3 mol Sb b. 0.0355 mol Ba c. 43.9 mol Xe d. 1.3 mol W
-
Which statements are consistent with Rutherfords nuclear theory as it was originally stated? Why? a. The volume of an atom is mostly empty space. b. The nucleus of an atom is small compared to the...
-
Design a fast LOT-based filter bank with at least eight sub-bands.
-
Prove the relationship in Equation (9.210). CC2=CC = 0, (9.210)
-
Show that the relations in Equations (9.256)-(9.258) are valid. E(z) = [C3+2 (I - 3)]4. (9.256)

Study smarter with the SolutionInn App


