(a) Predict the UV maximum for the reactant and product of the following photochemical reaction: CH3,...
Fantastic news! We've Found the answer you've been seeking!
Question:
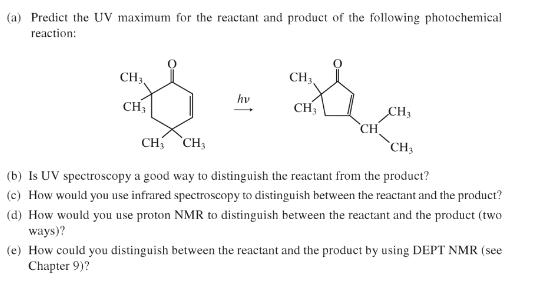
Transcribed Image Text:
(a) Predict the UV maximum for the reactant and product of the following photochemical reaction: CH3, CH3 hv CH; CH; CH3 CH CH CH3 `CH3 (b) Is UV spectroscopy a good way to distinguish the reactant from the product? (c) How would you use infrared spectroscopy to distinguish between the reactant and the product? (d) How would you use proton NMR to distinguish between the reactant and the product (two ways)? (e) How could you distinguish between the reactant and the product by using DEPT NMR (see Chapter 9)? (a) Predict the UV maximum for the reactant and product of the following photochemical reaction: CH3, CH3 hv CH; CH; CH3 CH CH CH3 `CH3 (b) Is UV spectroscopy a good way to distinguish the reactant from the product? (c) How would you use infrared spectroscopy to distinguish between the reactant and the product? (d) How would you use proton NMR to distinguish between the reactant and the product (two ways)? (e) How could you distinguish between the reactant and the product by using DEPT NMR (see Chapter 9)?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
How would you use infrared spectroscopy to distinguish between the following pairs of constitutionalisomers? (a) CH3C=CCH3 and CHCH2C3CH () CHH CHCCH2HH2 and (c) H CH3CH2CHO and
-
How would you use 1 H NMR spectroscopy to distinguish between the following compounds? (a) (b) (c) (d) (e) (f) . . . CI
-
How could you use infrared spectroscopy to distinguish between the following pairs of isomers? (a) HC CCH2NH2 and CH3CH2C N (b) CH3COCH3 and CH3CH2CHO
-
17. [1.1/2 Points] DETAILS PREVIOUS ANSWERS MY NOTES ASK YOUR TEACHER Write the composite function in the form f(g(x)). [Identify the inner function u-g(x) and the outer function y=f(u).) (Use...
-
Distinguish between a theory and a hypothesis.
-
The cables exert the forces shown on the pole. Assuming the pole is supported by a ball-and-socket joint at its base determine the components of reaction at A. The forces F1 and F2 lie in a...
-
Plaintiff contracted to install a boiler for defendant. After plaintiff had installed and tested the boiler, but before final payment to plaintiff had been made, defendant took custody of the boiler...
-
Assume that in an annual audit of Harlowe Inc. at December 31, 2014, you find the following transactions near the closing date. 1. A special machine, fabricated to order for a customer, was finished...
-
2024 2023 2022 Sales $ 78,000 Cost of goods sold 62,400 $ 70,000 60,900 $ 59,000 44,100 2021 $ 58,000 35,100 2020 $ 50,000 30,000 Required: Dollar amounts stated are in thousands. a. Compute trend...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Today is 15 April 2020. You are an audit manager of QUTPG Partners and are planning the audit of RST Co for the year ending 30 June 2020. The company is a manufacturer of digital devices and your...
-
Consider a circuit of two inductors, \(L_{1}\) and \(L_{2}\), in series. Prove that the equivalent inductance of the circuit is \(L_{\mathrm{eq}}=L_{1}+L_{2}\).
-
Go to the library or the Internet and, for a particular year, put together a data set of profits in agriculture in different geographical units of your state or country (e.g., counties of a U.S....
-
The Borda Count is a common way of making a choice among more than two alternatives. As defined in the text, each member of society assigns a rank to the social alternatives \((1,2, \ldots)\), with 1...
-
Consider a circuit of two capacitors, \(C_{1}\) and \(C_{2}\), in parallel. Prove that the equivalent capacitance of the circuit is \(C_{\mathrm{eq}}=C_{1}+C_{2}\).
-
The Bode plot of a first-order system indicates that the corner frequency is \(2.25 \mathrm{rad} / \mathrm{sec}\) and the approximate low-frequency logarithmic magnitude is \(-23.10 \mathrm{~dB}\)....
-
Why do organizations utilize economic principles to guide strategic short-term and long-term decision making?
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
The Wohl degradation, an alternative to the Ruff degradation, is nearly the reverse of the Kiliani-Fischer synthesis. The aldose carbonyl group is converted to the oxime, which is dehydrated by...
-
Predict the products formed by periodic acid cleavage of the following diols. (a) CH3CH(OH)CH(OH)CH3 (b) (c) (d) CHAOH OH OH Ph-CCH OH)CH,CH CH HO HO
-
A graduate student tried to make o-fluorophenylmagnesium bromide by adding magnesium to an ether solution of o-fluorobromobenzene. After obtaining puzzling results with this reaction, she repeated...
-
Professor Ray C. Fair's voting model was introduced in Exercise 2.23. He builds models that explain and predict the U.S. presidential elections. See his website at...
-
In the STAR experiment (Section 7.5.3), children were randomly assigned within schools into three types of classes: small classes with 13-17 students, regular-sized classes with 22-25 students, and...
-
Many cities in California have passed Inclusionary Zoning policies (also known as below-market housing mandates) as an attempt to make housing more affordable. These policies require developers to...

Study smarter with the SolutionInn App


