A protein sample complex consists of two proteins, a smaller protein, X, and a larger protein,...
Fantastic news! We've Found the answer you've been seeking!
Question:

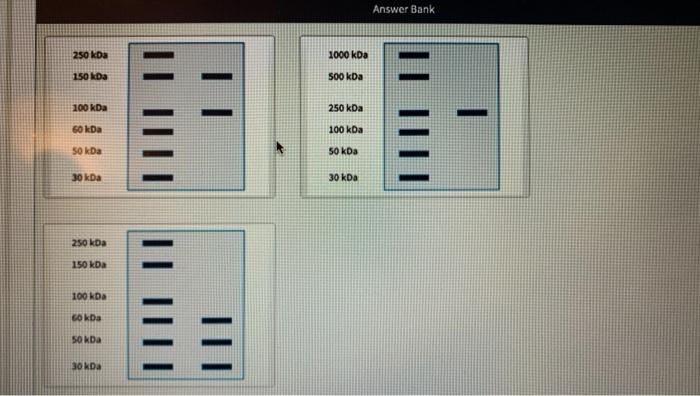

Transcribed Image Text:
A protein sample complex consists of two proteins, a smaller protein, X, and a larger protein, Y. Protein X is composed of two polypeptide chains linked by disulfide bonds. Protein Y is composed of three polypeptide chains linked by disulfide bonds. The complex is analyzed by native PAGE, reducing SDS-PAGE and non-reducing SDS-PAGE. Native PAGE does not include sodium dodecyl sulfate, or SDS. Reducing SDS-PAGE uses both SDS and a reducing agent in the buffer. Non-reducing SDS-PAGE uses SDS, but no reducing agent. Match each gel pattern to an experimental method. The first lane in each gel is a molecular weight ladder. Native PAGE Non-reducing SDS-PAGE Reducing SDS-PAGE Answer Bank Answer Bank 250 kDa 1000 kDa 150 kDa 500 kDa 100 kDa 250 kDa %3 60 kDa 100 kDa 50 kDa 50 kDa 30 kDa 30 kDa 250 kDa 150 kDa 100 kDa 60 kDa 50 kDa 30 kDa What are the molecular weights of the protein X polypeptides? What are the molecular weights of the protein Y polypeptides? A protein sample complex consists of two proteins, a smaller protein, X, and a larger protein, Y. Protein X is composed of two polypeptide chains linked by disulfide bonds. Protein Y is composed of three polypeptide chains linked by disulfide bonds. The complex is analyzed by native PAGE, reducing SDS-PAGE and non-reducing SDS-PAGE. Native PAGE does not include sodium dodecyl sulfate, or SDS. Reducing SDS-PAGE uses both SDS and a reducing agent in the buffer. Non-reducing SDS-PAGE uses SDS, but no reducing agent. Match each gel pattern to an experimental method. The first lane in each gel is a molecular weight ladder. Native PAGE Non-reducing SDS-PAGE Reducing SDS-PAGE Answer Bank Answer Bank 250 kDa 1000 kDa 150 kDa 500 kDa 100 kDa 250 kDa %3 60 kDa 100 kDa 50 kDa 50 kDa 30 kDa 30 kDa 250 kDa 150 kDa 100 kDa 60 kDa 50 kDa 30 kDa What are the molecular weights of the protein X polypeptides? What are the molecular weights of the protein Y polypeptides?
Expert Answer:
Related Book For 

Mathematical Applications for the Management Life and Social Sciences
ISBN: 978-1305108042
11th edition
Authors: Ronald J. Harshbarger, James J. Reynolds
Posted Date:
Students also viewed these chemical engineering questions
-
A 25.0-mL sample of sodium sulfate solution was analyzed by adding an excess of barium chloride solution to produce barium sulfate crystals, which were filtered from the solution. Na2SO4(aq) +...
-
Sodium triacetoxyborohydride, NaBH(OAc)3 is a mild reducing agent that reduces aldehydes much more quickly than ketones. It can be used to reduce aldehydes in the presence of ketones, such as in the...
-
A mixture contained aluminum sulfate and sodium sulfate. A sample of the mixture weighing 3.458 g was dissolved in water and treated with sodium hydroxide solution to yield a precipitate. The...
-
Explain the Mechanism & routes of administration of SEDDS (Self emulsifying drug delivery system).
-
Leo runs a bicycle repair shop. He recently exam-ined information on wage and employment levels and noted that he employs the same number of workers today that he employed in 2009. How-ever, wages...
-
Odessa, Inc., reports the following information concerning operations for the most recent month: Master Budget (based on budgeted 550 units) $93,500 Sales revenue Less Manufacturing costs Direct...
-
Determine the bending strain energy in the beam due to the loading shown. \(E I\) is constant. Mo -- B 22 -- C
-
In an effort to stop the migration of many of the automobile manufacturing facilities from the Detroit area, Detroits city council is considering passing a statute that would give investment tax...
-
ABC Companys budgeted sales for June, July, and August are 1 4 , 6 0 0 , 1 8 , 6 0 0 , and 1 6 , 6 0 0 units respectively. The selling price for each unit is $ 1 2 . Based on past experience, ABC...
-
The founder of Frenza asks us to assist her in accounting and analysis of the corporations bonds, which have an annual contract rate of 8%. She wants to know the business and accounting implications...
-
What would be the outcome if a leader who was not performing, not communicating effectively, or building relationships were to change? What underlying causes for the leader's behavior, and what can...
-
(a) Succinctly explain the methods of computing national income of a country citing the necessary adjustments to be taken in each case. (15 marks) (b) What is the difference between gross domestic...
-
(a) Differentiate between accelerator principle of investment and investment multiplier. (4marks) (b) Explain the determinants of induced investment in an economy. (8marks) (c) Given C=a+bY, explain...
-
(a) Briefly argue the case for and against protectionism policy practiced by countries of the world. (6marks) (b) Explain the term "Balance of Payment" and briefly highlight its components. (4marks)...
-
QUESTION FIVE (a) Explain how the government, through the central bank can use both fiscal and monetary policies to influence the stability and performance of an economy. (10 marks) (b) Explain the...
-
(a) (b) (c) Succinctly explain the methods of computing national income of a country citing the necessary adjustments to be taken in each case. (15 marks) What is the difference between gross...
-
How does the artist's innovative use of mixed media and found objects challenge traditional notions of artistic materiality and process, pushing the boundaries of artistic experimentation and...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Assume that sales revenues for Continental Divide Mining can be modeled by R(t) = 0.031t2 + 0.776t + 0.179 where t is the number of years past 2003. (a) Use the function to determine the year in...
-
By using Social Security Administration data for selected years from 2012 and projected to 2050, the U.S. consumer price index (CPI) can be modeled by the function C(t) = 92.7e0.0271t where t is the...
-
If f (x, y) = (x3 + 2y2)3, find f/x and f/y.
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]
-
Calculate the residual enthalpy and entropy for carbon dioxide at \(393 \mathrm{~K}\) and \(12 \mathrm{MPa}\) using any equation of state.

Study smarter with the SolutionInn App



