Question: A student performed three aqueous layer titrations in Part 1 and they reported the following measurements: Final Burette Reading Initial Burette Reading Volume of
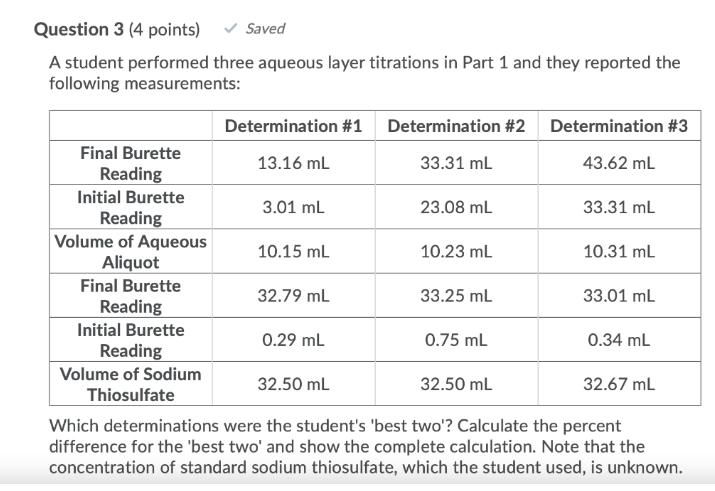
A student performed three aqueous layer titrations in Part 1 and they reported the following measurements: Final Burette Reading Initial Burette Reading Volume of Aqueous Aliquot Final Burette Reading Initial Burette Reading Volume of Sodium Thiosulfate Saved Determination #1 Determination #2 Determination #3 13.16 mL 33.31 mL 43.62 mL 33.31 mL 3.01 mL 10.15 mL 32.79 mL 0.29 mL 23.08 mL 10.23 mL 33.25 mL 0.75 mL 32.50 mL 10.31 mL 33.01 mL 0.34 mL 32.50 mL Which determinations were the student's 'best two'? Calculate the percent difference for the 'best two' and show the complete calculation. Note that the concentration of standard sodium thiosulfate, which the student used, is unknown. 32.67 ML
Step by Step Solution
3.45 Rating (152 Votes )
There are 3 Steps involved in it
The detailed ... View full answer

Get step-by-step solutions from verified subject matter experts


