a) What kind(s) of intermolecular forces exist in CHCl(1)? Select all that apply. dipole-dipole interactions hydrogen...
Fantastic news! We've Found the answer you've been seeking!
Question:
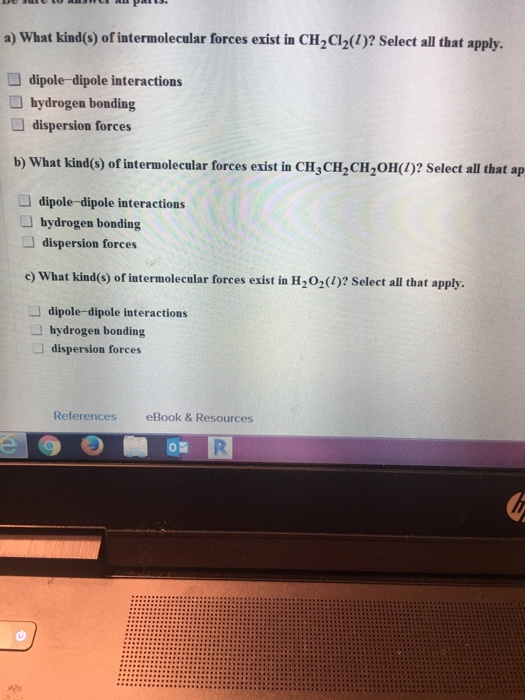
Transcribed Image Text:
a) What kind(s) of intermolecular forces exist in CH₂Cl₂(1)? Select all that apply. dipole-dipole interactions hydrogen bonding dispersion forces b) What kind(s) of intermolecular forces exist in CH3CH₂CH₂OH(1)? Select all that ap dipole-dipole interactions hydrogen bonding dispersion forces D コロ c) What kind(s) of intermolecular forces exist in H₂O₂(1)? Select all that apply. 77 dipole-dipole interactions hydrogen bonding dispersion forces References eBook & Resources R O a) What kind(s) of intermolecular forces exist in CH₂Cl₂(1)? Select all that apply. dipole-dipole interactions hydrogen bonding dispersion forces b) What kind(s) of intermolecular forces exist in CH3CH₂CH₂OH(1)? Select all that ap dipole-dipole interactions hydrogen bonding dispersion forces D コロ c) What kind(s) of intermolecular forces exist in H₂O₂(1)? Select all that apply. 77 dipole-dipole interactions hydrogen bonding dispersion forces References eBook & Resources R O
Expert Answer:
Answer rating: 100% (QA)
Dispersion Forces or London Dispersion Forces are the force of attraction between all molecules It ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Select all that apply to the Diamond Problem: C++ Compilers are equipped to handle this automatically with no further work from the programmer It can only occur when in the inheritance chain of an...
-
What kind of leader does the R & D team need, seeing as they are described as capable and eager? What leadership theory would this leader be applying and what would he or she need to provide for this...
-
Hydrogen bonding is a special case of very strong dipole dipole interactions possible among only certain atoms. What atoms in addition to hydrogen are necessary for hydrogen bonding? How does the...
-
In Problems 4164, graph each function using the techniques of shifting, compressing, stretching, and/or reflecting. Start with the graph of the basic function (for example, y = x 2 ) and show all the...
-
Jupiter Corporation manufactures home security devices. During 20x4, 1,000,000 units were completed and transferred to finished-goods inventory. On December 31, 20x4, there were 310,000 units in work...
-
Oriental Airlines is considering a capital expansion in which it will purchase new aircraft for its Pacific runs. Oriental is looking at the purchase of Boeing B797s, Airbus A450s, and Lockheed...
-
Three laser beams have wavelengths \(\lambda_{1}=400 \mathrm{~nm}, \lambda_{2}=\) \(600 \mathrm{~nm}\), and \(\lambda_{3}=800 \mathrm{~nm}\). The power of each laser beam is \(1 \mathrm{~W}\). a....
-
Sarah Stoner sells handmade jewelry that she designs herself. The items arent expensive, and she has a loyal following in her town and the surrounding area. Sarah incurred the following unit costs to...
-
How do demographic stochasticity, environmental variation, and dispersal dynamics interact to shape the spatial and temporal dynamics of populations within heterogeneous landscapes?
-
Go to the websites for project management tools (besides Microsoft Project), such as Apptivo (www.apptivo.com), GanttProject (www.ganttproject.biz), Gantter (www.gantter.com), and smartsheet...
-
How can motivation techniques can be applied to change management. Provide three (3) examples from the healthcare system and Human Resource Department. Provide a company's motivation style...
-
Given that f(x) = 3x + 5, find 2f(2) +5f(-6). 2f(2) +5f(-6) =
-
In what document would you most likely find information that states an employee must follow all local, state, and federal laws? Non - disparagement clause Code of conduct Non - solicitation clause...
-
9) About 40 TeraWatts is coming out of the Earth surface. On average, how many Joules of heat comes out of each square meter of Earth's surface in a day? Assume that Earth is a perfect sphere with a...
-
At which stage of the new product development process is a physical version of the market offering manufactured? A . Idea generation B . Test marketing C . Marketing strategy development D . Concept...
-
Problem 5 What is the charge (magnitude and sign) of a 2 gram particle if it is stationary in a downward electric field of magnitude 650 [N/C]? E = (650 [N/C])(-) FE F = (mg) (-y) 8
-
Define Organisational Behaviour. Explain Autocratic & custodial models of OB.
-
Study the pictures/images below. Obviously these was focus on LT sociology, anthropology and poltical science. Try to do some analysis by finding clues that are synonymous with the main concepts....
-
The partial pressures of an equilibrium mixture of N2O4(g) and NO2(g) are PN2O4 = 0.34 atm and PNO2 = 1.20 atm at a certain temperature. The volume of the container is doubled. Calculate the partial...
-
At a particular temperature, K = 3.75 for the reaction SO2(g) + NO2(g) SO3(g) + NO(g) If all four gases had initial concentrations of 0.800 M, calculate the equilibrium concentrations of the gases.
-
What tests could you perform to distinguish between the following pairs of compounds? a. b. c. d. CH3CH2NH2, CH3OCH3 CH3CH2CH2CH3, CH CHCH2CH3 CH,CH CH COOH CH,CH,CCH3
-
The required rate of return is 12%, and the expected rate of inflation over the life of the project is forecast to remain steady at 3%. Should you invest in this project?
-
For your answer to Problem 3.11, construct an argument as to why this selection process is useful for deciding which project is best. Now, construct a counterargument to criticize this choice. In...
-
Screening Model. Consider the following weighted criteria for assessing the viability of different charity event project proposals. Cost (3) Location (4) Team expertise (3) Celebrity endorsement...

Study smarter with the SolutionInn App


