(a) Which of the following half-reactions will oxidize Cu(s) to Cu+ under standard conditions? (i) Cl(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
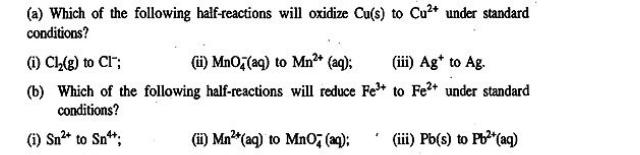
Transcribed Image Text:
(a) Which of the following half-reactions will oxidize Cu(s) to Cu²+ under standard conditions? (i) Cl₂(g) to CI™; (ii) MnO, (aq) to Mn²+ (aq); (iii) Ag* to Ag. (b) Which of the following half-reactions will reduce Fe³+ to Fe²+ under standard conditions? (i) Sn²+ to Sn¹+; (ii) Mn²+ (aq) to MnO2 (aq); (iii) Pb(s) to Pb²+ (aq) (a) Which of the following half-reactions will oxidize Cu(s) to Cu²+ under standard conditions? (i) Cl₂(g) to CI™; (ii) MnO, (aq) to Mn²+ (aq); (iii) Ag* to Ag. (b) Which of the following half-reactions will reduce Fe³+ to Fe²+ under standard conditions? (i) Sn²+ to Sn¹+; (ii) Mn²+ (aq) to MnO2 (aq); (iii) Pb(s) to Pb²+ (aq)
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The following half reactions play important roles in metabolism. 1/2 O2 1 2H+ + 2e H2O NADH + H+ NAD+ + 2H+ + 2e- Which of these two is a half reaction of oxidation? Which one is a half reaction of...
-
Which of the following reactions occurs most rapidly? Why? a. b. c. Br - H20 C(CH3)3 C(CH3)a (CH)a C(CH3)3 Br - H20 CH33 C(CH3)3
-
Which of the following reactions and decays are possible? For those forbidden, explain what laws are violated. (a) + p n + 0 (b) + + p n + 0 (c) + + p p + e0 (d) p e+ + vp (e) + e+ + vp (f) p n + e...
-
You have $300,000 saved for retirement. Your account earns 6% interest. How much will you be able to pull out each month, if you want to be able to take withdrawals for 20 years? $ Get help: Video
-
Submit either a one- to three-page questionnaire or questions for a 20- to 30-minute interview AND the information listed below for the method you choose. Questionnaire 1. Purpose(s), goal(s). 2....
-
The approval process for new drugs, if governed by economic thinking, should set stringency standards so that the __________ equals the ___________. a. Total cost; total benefit b. Average cost;...
-
You need to lower a \(45-\mathrm{kg}\) safe from a window to the bed of a \(1800-\mathrm{kg}\) truck. You have a rope that is just long enough, but it will support no more than \(42 \mathrm{~kg}\)....
-
The Stopdecay Company sells an electric toothbrush for $25. Its sales have averaged 8,000 units per month over the past year. Recently, its closest competitor, Decayfighter, reduced the price of its...
-
You long one Russell 1000 index future contract (the multiplier is $1000 for one contract). Assume the initial margin requirement is $200,000, and the maintenance margin requirement is $150,000, will...
-
The Picking, Packing, and Shipping process uses 4 different teams: Picking team - gets items from the shelves and sends to Packing Group A and Packing Group B Packing Group A - packs items from the...
-
A community has a 125-year-old bridge, and a project has been created to restore the bridge and give it historical status. However, the project is in need of funding and support. The bridge is...
-
Determine power at one load (any R, L or C component in the circuit) using Fourier Series analysis. 1000w w2s2+10w2s+100w2+s4 + 10s + 100s V(s)= I(s) = s2+2+10+8+ 0.018 10w
-
The number of units of output that an acre of land will produce increases, ceteris paribus. How will this change in productivity affect demand for the land?
-
1. How do you cite a source if you can't find the author's name? 2. Describe (or imagine) a time when somebody used your idea: a. Were you pleased or upset? b. Did they use your idea without asking...
-
Describe why feasibility studies are necessary. How is a SWOT analysis used to develop a marketing plan Explain why it is necessary to segment. What are the elements that make up segmentation How...
-
Explain how Base Erosion Profit Shifting (BEPS) initiative is changing the dynamics of Double Tax Agreements (DTA) and Transfer Pricing regimes around the world and how these will impact on Fiji's...
-
One Energy, Inc. manufactures and sells portable solar generators. Its market is rapidly growing due to declining cost of solar panel technology and increased awareness of the environmental benefits...
-
In muscle tissue, the ratio of phosphorylase a to phosphorylase b determines the rate of conversion of glycogen to glucose 1phosphate. Classify how each event affects the rate of glycogen breakdown...
-
For the equilibrium at 400 K, Kc = 7.0. If 0.25 mol of Br2 and 0.55 mol of Cl2 are introduced into a 3.0-L container at 400 K, what will be the equilibrium concentrations of Br2, Cl2, and BrCl?
-
If the equilibrium constant for a two-electron redox reaction at 298 K is 1.5 10-4, calculate the corresponding G and Ered.
-
Consider the following three reactions: (a) For each of the reactions, use data in Appendix C to calculate H, G, and S at 25C. (b) Which of these reactions are spontaneous under standard conditions...
-
An object traveling at a constant velocity vo experiences a constant acceleration in the same direction for a period of time t. Then an acceleration of equal magnitude is experienced in the opposite...
-
Can the displacement of a persons trip be zero, yet the distance involved in the trip is nonzero? How about the reverse situation? Explain.
-
You are told that a person has walked 750 m. What can you safely say about the persons final position relative to the starting point?

Study smarter with the SolutionInn App



